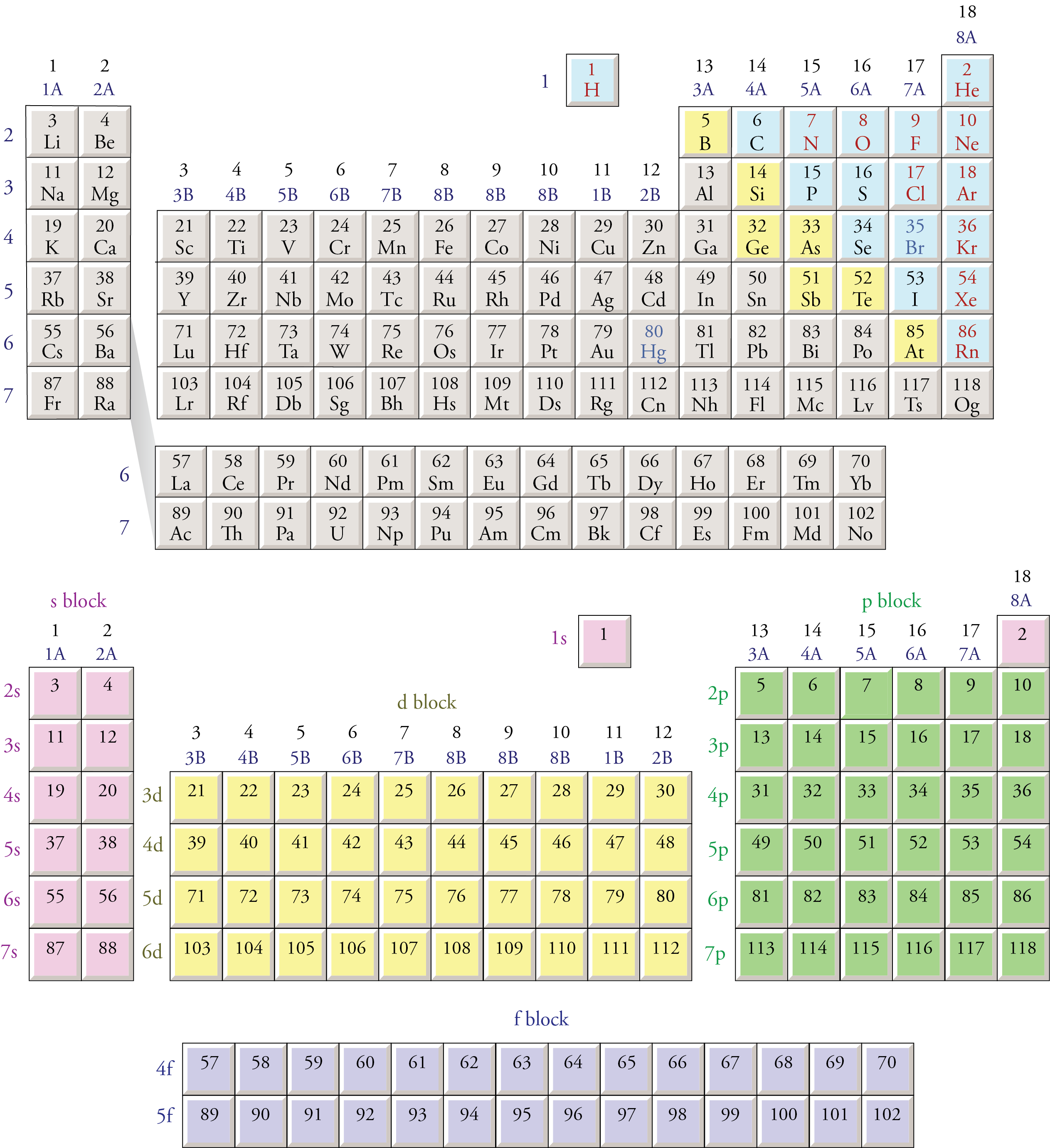
The Periodic Table And Orbitals is a fundamental concept in chemistry that helps us understand the structure and behavior of atoms. The periodic table organizes elements based on their atomic number, electron configuration, and recurring chemical properties. Orbitals, on the other hand, describe the regions where electrons are likely to be found around the nucleus. Understanding the relationship between the periodic table and orbitals is crucial for grasping the principles of atomic structure and chemical bonding.
The Structure of the Periodic Table
The periodic table is arranged in rows and columns. The rows are called periods, and the columns are called groups. Each element in the periodic table has a unique atomic number, which corresponds to the number of protons in its nucleus. The elements are organized in a way that reflects their electron configurations, which in turn determine their chemical properties.
The periodic table is divided into several blocks based on the type of orbital being filled with electrons:
- s-block: Elements in groups 1 and 2, where the outermost electrons are in s-orbitals.
- p-block: Elements in groups 13 to 18, where the outermost electrons are in p-orbitals.
- d-block: Transition metals in groups 3 to 12, where the outermost electrons are in d-orbitals.
- f-block: Lanthanides and actinides, where the outermost electrons are in f-orbitals.
Understanding Orbitals
Orbitals are mathematical functions that describe the wave-like behavior of electrons in an atom. Each orbital can hold a maximum of two electrons, which must have opposite spins. The four types of orbitals are s, p, d, and f, each with a distinct shape and energy level.
The s-orbital is spherical and can hold up to two electrons. The p-orbitals are dumbbell-shaped and come in three orientations (px, py, pz), each capable of holding two electrons. The d-orbitals have more complex shapes and come in five orientations (dxy, dxz, dyz, dx2-y2, dz2), each holding two electrons. The f-orbitals are even more complex and come in seven orientations, each holding two electrons.
The Relationship Between the Periodic Table and Orbitals
The arrangement of elements in the periodic table directly reflects the filling of orbitals with electrons. As you move from left to right across a period, electrons are added to the same energy level but different orbitals. For example, in the second period, electrons fill the 2s and 2p orbitals:
| Element | Atomic Number | Electron Configuration |
|---|---|---|
| Lithium (Li) | 3 | 1s22s1 |
| Beryllium (Be) | 4 | 1s22s2 |
| Boron (B) | 5 | 1s22s22p1 |
| Carbon (C) | 6 | 1s22s22p2 |
| Nitrogen (N) | 7 | 1s22s22p3 |
| Oxygen (O) | 8 | 1s22s22p4 |
| Fluorine (F) | 9 | 1s22s22p5 |
| Neon (Ne) | 10 | 1s22s22p6 |
As you move down a group, electrons are added to the next higher energy level. For example, in group 1 (alkali metals), each element has one electron in its outermost s-orbital:
| Element | Atomic Number | Electron Configuration |
|---|---|---|
| Lithium (Li) | 3 | 1s22s1 |
| Sodium (Na) | 11 | 1s22s22p63s1 |
| Potassium (K) | 19 | 1s22s22p63s23p64s1 |
This pattern continues for other groups and periods, illustrating how the periodic table is a visual representation of the filling of orbitals with electrons.
Periodic Trends and Orbitals
The periodic table also helps us understand various periodic trends, which are patterns in the properties of elements based on their position in the table. These trends are directly related to the electron configurations and orbitals:
- Atomic Radius: Generally decreases from left to right across a period and increases down a group. This is due to the increasing number of electron shells and the shielding effect of inner electrons.
- Ionization Energy: Generally increases from left to right across a period and decreases down a group. This is because it becomes harder to remove an electron from a smaller, more tightly bound atom.
- Electronegativity: Generally increases from left to right across a period and decreases down a group. This is due to the increasing attraction between the nucleus and the outermost electrons.
- Electron Affinity: Generally increases from left to right across a period and decreases down a group. This is because smaller atoms can more easily accommodate additional electrons.
These trends are crucial for understanding the chemical behavior of elements and their compounds.
🔍 Note: The trends mentioned above are generalizations and may have exceptions due to the unique electronic structures of certain elements.
Chemical Bonding and Orbitals
Understanding the Periodic Table And Orbitals is also essential for comprehending chemical bonding. Chemical bonds form when atoms share or transfer electrons to achieve a stable electron configuration. The type of bond formed depends on the orbitals involved:
- Ionic Bonds: Form when one atom donates electrons to another, resulting in positively and negatively charged ions. This typically occurs between metals (which have low ionization energies) and nonmetals (which have high electron affinities).
- Covalent Bonds: Form when atoms share electrons to fill their outermost orbitals. This typically occurs between nonmetals, which have similar electronegativities.
- Metallic Bonds: Form when metal atoms share a "sea" of delocalized electrons, resulting in a lattice of positive ions. This gives metals their characteristic properties, such as conductivity and malleability.
In covalent bonding, the type of orbital overlap determines the bond's strength and directionality. For example, sigma (σ) bonds form when orbitals overlap head-to-head, while pi (π) bonds form when orbitals overlap side-to-side. These bonds can be further classified based on the orbitals involved, such as sp, sp2, sp3, and sp3d hybrid orbitals.
Applications of the Periodic Table and Orbitals
The understanding of the Periodic Table And Orbitals has numerous applications in various fields, including:
- Material Science: The periodic table helps in the development of new materials with desired properties by predicting the behavior of elements and their compounds.
- Pharmaceuticals: The understanding of electron configurations and orbitals is crucial for designing drugs that interact with specific biological targets.
- Environmental Science: The periodic table aids in understanding the behavior of pollutants and their impact on the environment.
- Energy: The knowledge of atomic structure and orbitals is essential for developing new energy sources, such as nuclear fusion and advanced batteries.
In conclusion, the Periodic Table And Orbitals are fundamental concepts in chemistry that provide a framework for understanding the structure and behavior of atoms. By studying the periodic table and orbitals, we can predict the properties of elements, understand chemical bonding, and apply this knowledge to various fields. The periodic table’s organization based on electron configurations and orbitals highlights the underlying patterns in the chemical world, making it an invaluable tool for chemists and scientists alike.
Related Terms:
- periodic table with orbital levels
- periodic table with orbital labels
- orbital naming chart
- orbital chart on periodic table
- orbitals chemistry periodic table
- orbital periodic tabel