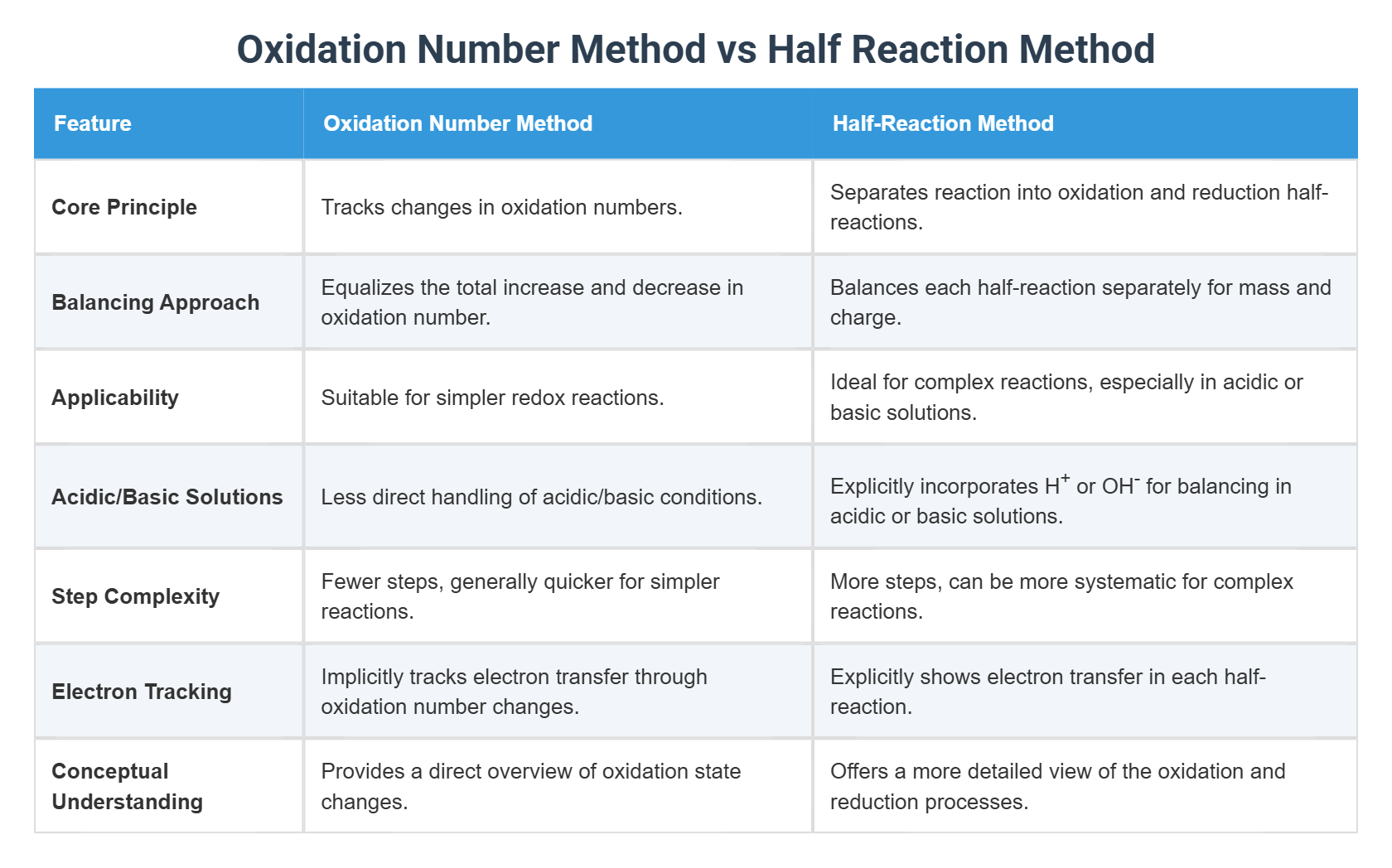
Understanding the fundamentals of redox reactions is crucial for anyone delving into the world of chemistry. At the heart of these reactions lies the oxidation half reaction, a process that involves the loss of electrons by a substance. This blog post will explore the intricacies of oxidation half reactions, their significance, and how they are balanced and identified in chemical equations.
What is an Oxidation Half Reaction?
An oxidation half reaction is a part of a redox reaction where a substance loses electrons. This process is known as oxidation. In a redox reaction, both oxidation and reduction occur simultaneously. The substance that undergoes oxidation is said to be oxidized, while the substance that gains electrons is reduced. The oxidation half reaction is essential for understanding the overall redox process and balancing chemical equations.
Identifying Oxidation Half Reactions
To identify an oxidation half reaction, you need to look for changes in the oxidation states of the elements involved. The oxidation state of an element increases during oxidation. Here are the steps to identify an oxidation half reaction:
- Determine the oxidation states of all elements in the reactants and products.
- Identify which element’s oxidation state increases.
- Write the half reaction for the element that is being oxidized, showing the loss of electrons.
Balancing Oxidation Half Reactions
Balancing oxidation half reactions is a critical step in understanding redox chemistry. The process involves ensuring that the number of electrons lost in the oxidation half reaction matches the number of electrons gained in the reduction half reaction. Here are the steps to balance an oxidation half reaction:
- Write the unbalanced half reaction showing the loss of electrons.
- Balance the atoms other than hydrogen and oxygen.
- Balance the oxygen atoms by adding water molecules (H2O) to the side that needs oxygen.
- Balance the hydrogen atoms by adding hydrogen ions (H+) to the side that needs hydrogen.
- Balance the charge by adding electrons (e-) to the side that needs a negative charge.
💡 Note: In acidic solutions, hydrogen ions (H+) are added to balance hydrogen atoms. In basic solutions, hydroxide ions (OH-) are added instead.
Examples of Oxidation Half Reactions
Let’s look at a few examples to illustrate the process of identifying and balancing oxidation half reactions.
Example 1: Oxidation of Iron
Consider the oxidation of iron (Fe) to iron(III) oxide (Fe2O3). The unbalanced half reaction for the oxidation of iron is:
Fe → Fe3+ + 3e-
This half reaction shows that iron loses three electrons to form iron(III) ions.
Example 2: Oxidation of Sulfur
Another example is the oxidation of sulfur (S) to sulfate ions (SO42-). The unbalanced half reaction is:
S → SO42-
To balance this half reaction, we follow the steps outlined earlier:
- Balance the sulfur atoms: S → SO42-
- Balance the oxygen atoms by adding water molecules: S + 4H2O → SO42-
- Balance the hydrogen atoms by adding hydrogen ions: S + 4H2O → SO42- + 8H+
- Balance the charge by adding electrons: S + 4H2O → SO42- + 8H+ + 6e-
The balanced oxidation half reaction is:
S + 4H2O → SO42- + 8H+ + 6e-
Applications of Oxidation Half Reactions
Oxidation half reactions have numerous applications in various fields, including:
- Batteries and Fuel Cells: Oxidation half reactions are crucial in the functioning of batteries and fuel cells, where chemical energy is converted into electrical energy.
- Corrosion: Understanding oxidation half reactions helps in studying and preventing corrosion, which is the oxidation of metals.
- Biochemistry: In biological systems, oxidation half reactions play a vital role in metabolic processes, such as respiration and photosynthesis.
- Environmental Chemistry: Oxidation half reactions are involved in the degradation of pollutants and the cycling of nutrients in the environment.
Common Mistakes to Avoid
When working with oxidation half reactions, it’s essential to avoid common mistakes that can lead to incorrect balancing and understanding. Some of these mistakes include:
- Not Balancing Atoms Properly: Ensure that all atoms, except hydrogen and oxygen, are balanced before adding water molecules.
- Ignoring Charge Balance: Always balance the charge by adding the correct number of electrons.
- Confusing Oxidation and Reduction: Remember that oxidation involves the loss of electrons, while reduction involves the gain of electrons.
💡 Note: Double-check your work to ensure that the number of electrons lost in the oxidation half reaction matches the number of electrons gained in the reduction half reaction.
Advanced Topics in Oxidation Half Reactions
For those interested in delving deeper into the subject, there are several advanced topics to explore:
- Electrochemistry: Study the relationship between electrical energy and chemical reactions, focusing on oxidation and reduction processes.
- Redox Titrations: Learn about titration methods that involve redox reactions, such as permanganate titrations.
- Nernst Equation: Understand the thermodynamic aspects of redox reactions using the Nernst equation, which relates the cell potential to the standard electrode potential and the concentrations of reactants and products.
These advanced topics provide a deeper understanding of oxidation half reactions and their applications in various scientific and industrial fields.
Conclusion
Oxidation half reactions are a fundamental concept in redox chemistry, involving the loss of electrons by a substance. Understanding how to identify and balance these reactions is crucial for studying redox processes and their applications. By following the steps outlined in this post, you can accurately identify and balance oxidation half reactions, gaining a deeper insight into the world of chemistry. Whether you’re studying corrosion, batteries, or biological processes, a solid grasp of oxidation half reactions will serve as a valuable foundation for your chemical knowledge.
Related Terms:
- oxidation half reaction table
- oxidation half reaction equation
- oxidation half reaction formula
- oxidation half reaction calculator
- oxidation half reaction meaning
- redox reaction