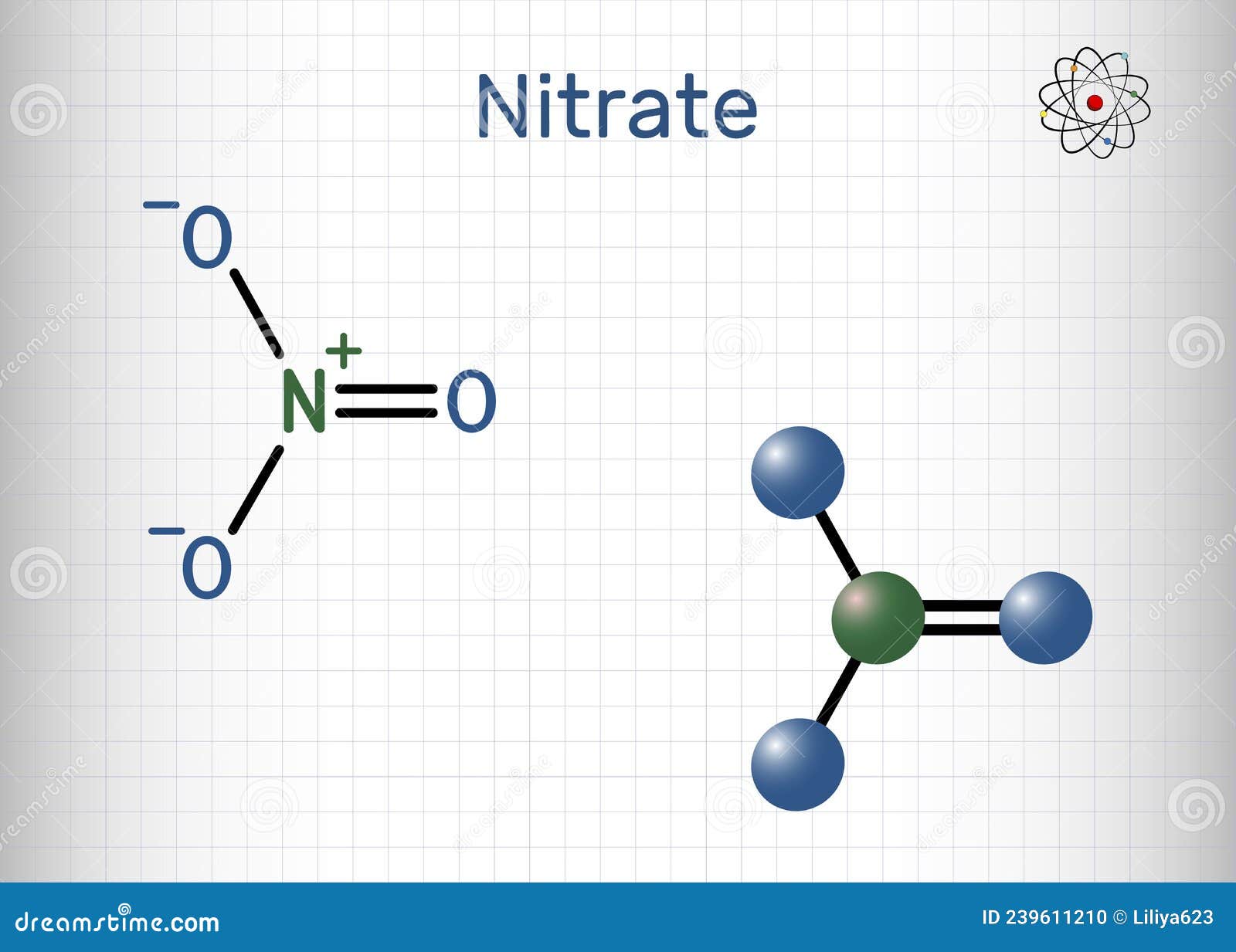
Understanding the Nitrate Anion Lewis Structure is fundamental for anyone studying chemistry, particularly those delving into the intricacies of molecular bonding and electron distribution. The nitrate anion, with the chemical formula NO₃⁻, is a polyatomic ion that plays a crucial role in various chemical reactions and biological processes. This blog post will explore the Nitrate Anion Lewis Structure, its properties, and its significance in chemistry.
Understanding the Nitrate Anion
The nitrate anion is composed of one nitrogen atom and three oxygen atoms. It carries a negative charge of -1, making it an essential component in many chemical compounds and reactions. The nitrate ion is commonly found in fertilizers, explosives, and even in the human body as a byproduct of metabolism.
The Lewis Structure of Nitrate Anion
The Nitrate Anion Lewis Structure provides a visual representation of how electrons are distributed among the atoms in the ion. To draw the Lewis structure, follow these steps:
- Determine the total number of valence electrons. For NO₃⁻, nitrogen has 5 valence electrons, each oxygen has 6 valence electrons, and there is an additional electron due to the negative charge. This gives a total of 24 valence electrons.
- Identify the central atom. In the nitrate ion, nitrogen (N) is the central atom.
- Connect the central atom to the surrounding atoms with single bonds. This uses 6 electrons (3 bonds × 2 electrons per bond).
- Distribute the remaining electrons to complete the octets of the surrounding atoms. Each oxygen atom needs 2 more electrons to complete its octet, using up 18 electrons in total.
- Place any remaining electrons on the central atom. In this case, there are no remaining electrons, so the nitrogen atom has a formal charge of +1.
- Adjust the bonds to minimize formal charges. Convert lone pairs on oxygen atoms into pi bonds with the central nitrogen atom to form double bonds. This results in a resonance structure where each oxygen atom has a double bond with the nitrogen atom.
Here is the final Nitrate Anion Lewis Structure:

Resonance Structures of Nitrate Anion
The nitrate ion exhibits resonance, meaning there are multiple valid Lewis structures that represent the same molecule. Each resonance structure contributes to the overall stability of the ion. The three resonance structures of the nitrate ion are:
- One oxygen atom has a double bond with the nitrogen atom, and the other two oxygen atoms have single bonds.
- Another oxygen atom has a double bond with the nitrogen atom, and the other two oxygen atoms have single bonds.
- The third oxygen atom has a double bond with the nitrogen atom, and the other two oxygen atoms have single bonds.
These resonance structures help to distribute the negative charge evenly across the ion, making it more stable.
Properties of the Nitrate Anion
The nitrate anion has several important properties that make it significant in various applications:
- Stability: The nitrate ion is relatively stable due to its resonance structures, which help to distribute the negative charge.
- Solubility: Nitrate salts are generally soluble in water, making them useful in fertilizers and other applications.
- Oxidizing Agent: The nitrate ion can act as an oxidizing agent in chemical reactions, accepting electrons from other substances.
- Biological Role: Nitrate is an essential nutrient for plants and is a key component in the nitrogen cycle.
Applications of the Nitrate Anion
The nitrate anion has a wide range of applications in various fields:
- Fertilizers: Nitrate is a common component in fertilizers due to its role as a nutrient for plants.
- Explosives: Nitrate salts, such as ammonium nitrate, are used in the production of explosives.
- Food Preservation: Sodium nitrate is used as a preservative in cured meats to inhibit bacterial growth.
- Industrial Processes: Nitrate is used in various industrial processes, including the production of dyes, pigments, and other chemicals.
Environmental Impact of Nitrate
While the nitrate anion has many beneficial applications, it also has environmental impacts that need to be considered:
- Water Pollution: Excessive use of nitrate fertilizers can lead to water pollution, particularly in groundwater and surface water bodies.
- Eutrophication: High levels of nitrate in water can cause eutrophication, leading to the overgrowth of algae and the depletion of oxygen, which can harm aquatic life.
- Health Risks: High levels of nitrate in drinking water can pose health risks, particularly to infants, as it can interfere with the oxygen-carrying capacity of blood.
It is essential to manage the use of nitrate compounds responsibly to minimize these environmental and health impacts.
📝 Note: Always follow local regulations and best practices when handling and disposing of nitrate compounds to ensure environmental safety.
Conclusion
The Nitrate Anion Lewis Structure provides valuable insights into the bonding and electron distribution within the nitrate ion. Understanding this structure is crucial for comprehending the properties and applications of nitrate in various fields. From its role as a nutrient in fertilizers to its use in explosives and food preservation, the nitrate anion plays a significant part in both natural and industrial processes. However, it is also important to be aware of the environmental and health impacts associated with nitrate compounds and to manage their use responsibly.
Related Terms:
- best lewis structure for no3
- nitrate lewis diagram
- three lewis structures for no3
- lewis structure for no3 1
- nitrate ion diagram
- draw lewis structure for no3