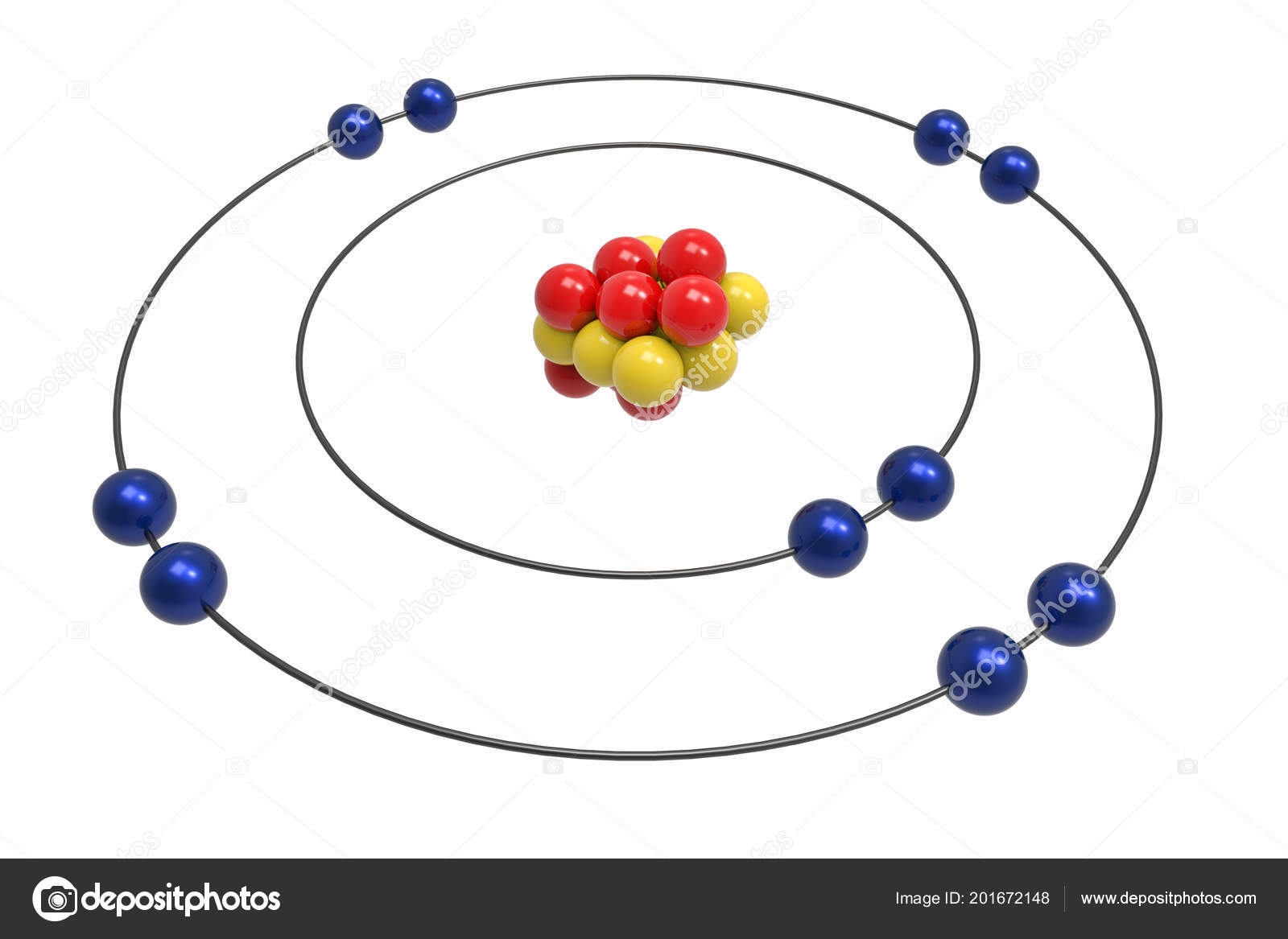
Understanding the structure of atoms is fundamental to grasping the principles of chemistry and physics. One of the most iconic representations of atomic structure is the Bohr model, which provides a simplified yet effective way to visualize the arrangement of electrons around the nucleus. Among the elements, neon stands out due to its unique properties and stability. Exploring the Neon Element Bohr Model offers insights into the electronic configuration and stability of this noble gas.
The Bohr Model: A Brief Overview
The Bohr model, proposed by Niels Bohr in 1913, revolutionized the understanding of atomic structure. It introduced the concept of discrete energy levels, or shells, where electrons orbit the nucleus at fixed distances. Each shell can hold a specific number of electrons, and the outermost shell determines the chemical properties of an element.
Neon: The Noble Gas
Neon is a chemical element with the symbol Ne and atomic number 10. It is a colorless, odorless, inert monatomic gas that is part of the noble gas group on the periodic table. Neon is known for its use in advertising signs and high-voltage indicators due to its distinctive red-orange glow when electrically excited.
The Electronic Configuration of Neon
The electronic configuration of an element describes the arrangement of electrons in its atomic orbitals. For neon, the electronic configuration is 1s22s22p6. This means that neon has two electrons in the first shell (1s), two electrons in the second shell (2s), and six electrons in the second shell (2p).
The Neon Element Bohr Model
The Neon Element Bohr Model visually represents this electronic configuration. In the Bohr model, the nucleus is at the center, surrounded by concentric circles representing the energy levels or shells. For neon, there are two shells:
- The first shell (K-shell) contains 2 electrons.
- The second shell (L-shell) contains 8 electrons (2 in the 2s sub-shell and 6 in the 2p sub-shell).
This arrangement can be visualized as follows:
| Shell | Sub-shell | Number of Electrons |
|---|---|---|
| K-shell | 1s | 2 |
| L-shell | 2s | 2 |
| L-shell | 2p | 6 |
Stability of Neon
Neon’s stability is a result of its fully filled outer shell. In the Bohr model, the outermost shell (L-shell) is completely filled with 8 electrons, making neon a highly stable and non-reactive element. This stability is a characteristic of all noble gases, which have fully filled outer shells.
Applications of Neon
Neon’s unique properties make it valuable in various applications:
- Lighting: Neon is commonly used in neon signs and high-voltage indicators due to its bright red-orange glow when electrically excited.
- Cryogenics: Liquid neon is used as a cryogenic refrigerant in applications requiring temperatures below those achievable with liquid helium.
- Lasers: Neon is used in helium-neon lasers, which are commonly used in scientific research and medical applications.
Comparing Neon with Other Noble Gases
Neon is one of several noble gases, each with its unique properties. Here is a brief comparison:
| Element | Atomic Number | Electronic Configuration | Common Uses |
|---|---|---|---|
| Helium (He) | 2 | 1s2 | Balloons, cooling in MRI machines |
| Neon (Ne) | 10 | 1s22s22p6 | Neon signs, cryogenics, lasers |
| Argon (Ar) | 18 | 1s22s22p63s23p6 | Welding, light bulbs |
| Krypton (Kr) | 36 | 1s22s22p63s23p63d104s24p6 | Fluorescent lamps, lasers |
| Xenon (Xe) | 54 | 1s22s22p63s23p63d104s24p64d105s25p6 | Anesthesia, automotive headlights |
💡 Note: The electronic configurations listed above are simplified and do not include the detailed sub-shells for clarity.
Visualizing the Neon Element Bohr Model
To better understand the Neon Element Bohr Model, consider the following visual representation:

This diagram shows the nucleus at the center, with the first shell containing 2 electrons and the second shell containing 8 electrons. The second shell is divided into 2s and 2p sub-shells, with 2 electrons in the 2s sub-shell and 6 electrons in the 2p sub-shell.
Understanding the Neon Element Bohr Model provides a clear picture of neon’s atomic structure and stability. The fully filled outer shell explains why neon is inert and non-reactive, making it a valuable element in various applications. By studying the Bohr model, we gain insights into the fundamental principles of atomic structure and the behavior of elements.
Related Terms:
- bohr rutherford diagram
- neon bohr model diagram
- diagram of a neon atom
- electron shell diagram for neon
- neon energy level diagram
- neon atom bohr diagram