Understanding the molecular shape of phosphine (PH3) is crucial for chemists and researchers in various fields, including inorganic chemistry, materials science, and environmental studies. The molecular shape of PH3, or phosphine, plays a significant role in its chemical behavior and interactions. This post delves into the intricacies of the molecular shape of PH3, its implications, and how it influences various chemical processes.
Understanding the Molecular Shape of PH3
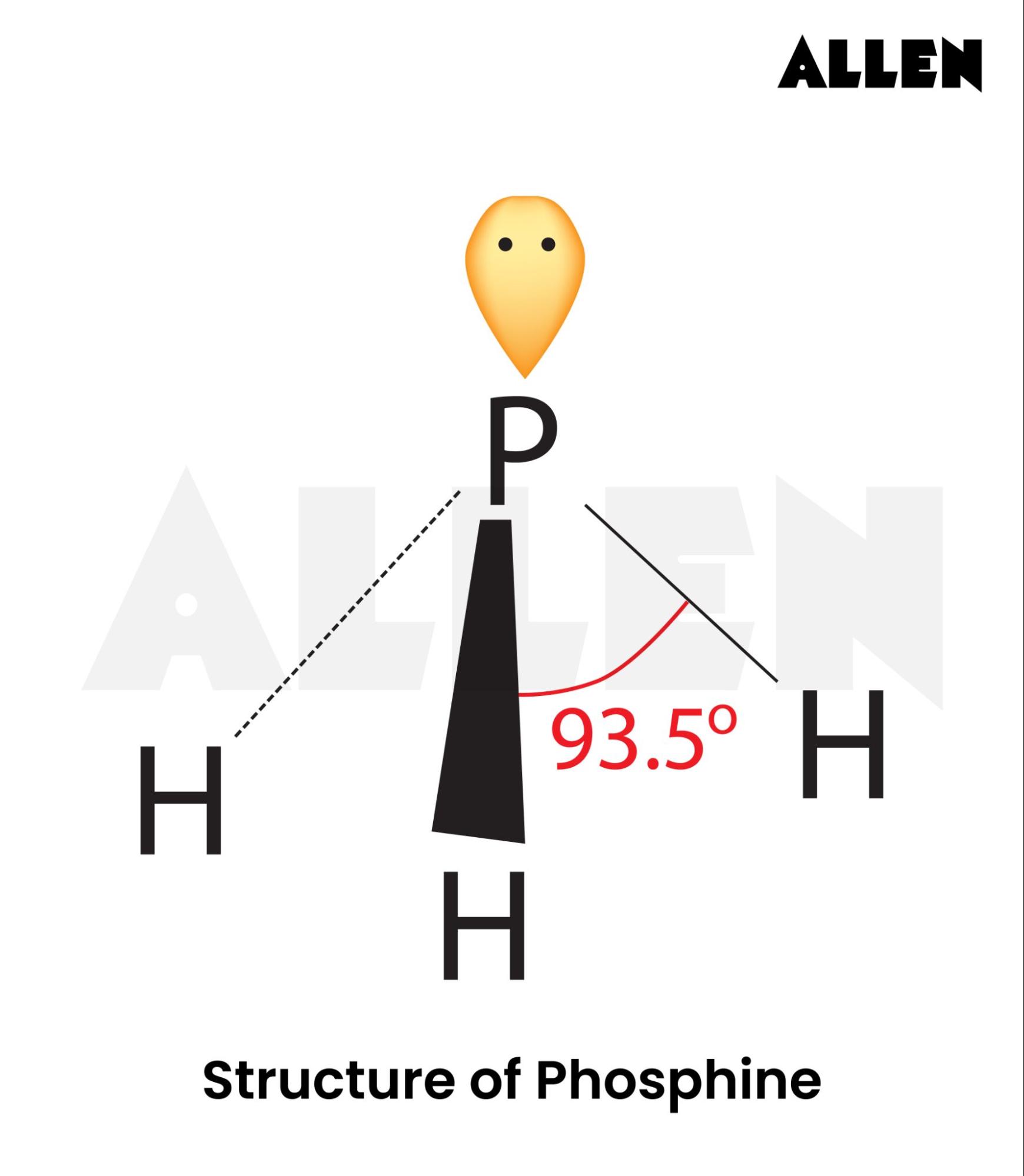
The molecular shape of PH3 is determined by the arrangement of its atoms and the electron pairs around the central phosphorus atom. Phosphine has a trigonal pyramidal shape, which is a result of the electron pair geometry and the lone pair of electrons on the phosphorus atom. This shape is crucial for understanding its reactivity and bonding properties.
To comprehend the molecular shape of PH3, it is essential to look at its electron pair geometry. The central phosphorus atom in PH3 has five valence electrons. It forms three single bonds with three hydrogen atoms, using three of its valence electrons. The remaining two electrons form a lone pair. According to the Valence Shell Electron Pair Repulsion (VSEPR) theory, the lone pair and the three bonding pairs repel each other, leading to a trigonal pyramidal shape.
The bond angles in PH3 are slightly less than the ideal tetrahedral angle of 109.5 degrees due to the presence of the lone pair. The lone pair repels the bonding pairs more strongly, causing the H-P-H bond angles to be approximately 93.5 degrees. This deviation from the ideal tetrahedral angle is a key characteristic of the molecular shape of PH3.
Factors Influencing the Molecular Shape of PH3
Several factors influence the molecular shape of PH3, including:
- Electron Pair Repulsion: The lone pair on the phosphorus atom repels the bonding pairs more strongly, leading to a distorted tetrahedral geometry.
- Hybridization: The phosphorus atom in PH3 undergoes sp3 hybridization, which contributes to the trigonal pyramidal shape.
- Steric Effects: The spatial arrangement of the atoms and electron pairs affects the overall molecular shape.
These factors collectively determine the unique molecular shape of PH3, which in turn affects its chemical properties and reactivity.
Implications of the Molecular Shape of PH3
The molecular shape of PH3 has several important implications in chemistry and related fields. Understanding these implications can help researchers predict the behavior of PH3 in various chemical reactions and applications.
One of the key implications is the reactivity of PH3. The lone pair on the phosphorus atom makes PH3 a good nucleophile, meaning it can donate electrons to form new bonds. This property is crucial in many chemical reactions, including substitution and addition reactions. The trigonal pyramidal shape allows PH3 to approach other molecules from different angles, facilitating various types of reactions.
Another important implication is the solubility of PH3. The molecular shape and the presence of the lone pair influence the intermolecular forces between PH3 molecules and other substances. PH3 is slightly soluble in water due to its ability to form hydrogen bonds with water molecules. This solubility is essential in environmental studies, where PH3 can interact with water bodies and affect aquatic ecosystems.
Additionally, the molecular shape of PH3 affects its use in materials science. PH3 is used as a precursor in the synthesis of various materials, including semiconductors and catalysts. The trigonal pyramidal shape allows PH3 to interact with other molecules in specific ways, leading to the formation of desired materials with unique properties.
Comparing the Molecular Shape of PH3 with Other Molecules
Comparing the molecular shape of PH3 with other molecules can provide insights into its unique properties and behavior. For example, ammonia (NH3) has a similar trigonal pyramidal shape due to the presence of a lone pair on the nitrogen atom. However, the bond angles in NH3 are slightly different from those in PH3 due to the differences in atomic size and electronegativity.
Another molecule to compare is methane (CH4), which has a tetrahedral shape. The absence of a lone pair in CH4 leads to a more symmetrical arrangement of the hydrogen atoms around the central carbon atom. This comparison highlights the role of lone pairs in determining molecular shape and reactivity.
Below is a table comparing the molecular shapes and properties of PH3, NH3, and CH4:
| Molecule | Molecular Shape | Bond Angles | Presence of Lone Pair |
|---|---|---|---|
| PH3 | Trigonal Pyramidal | Approximately 93.5 degrees | Yes |
| NH3 | Trigonal Pyramidal | Approximately 107 degrees | Yes |
| CH4 | Tetrahedral | 109.5 degrees | No |
📝 Note: The presence of a lone pair in PH3 and NH3 leads to a distorted tetrahedral geometry, while the absence of a lone pair in CH4 results in a perfect tetrahedral shape.
Applications of PH3 in Various Fields
The molecular shape of PH3 makes it a valuable compound in various fields, including chemistry, materials science, and environmental studies. Understanding its molecular shape and properties can help researchers develop new applications and improve existing ones.
In chemistry, PH3 is used as a reducing agent in organic synthesis. Its ability to donate electrons makes it an effective reducing agent in various reactions, including the reduction of aldehydes and ketones to alcohols. The trigonal pyramidal shape allows PH3 to approach other molecules from different angles, facilitating the reduction process.
In materials science, PH3 is used as a precursor in the synthesis of semiconductors and catalysts. The molecular shape of PH3 allows it to interact with other molecules in specific ways, leading to the formation of materials with unique properties. For example, PH3 can be used to synthesize phosphorous-doped silicon, which is used in the production of solar cells and other electronic devices.
In environmental studies, PH3 is studied for its role in atmospheric chemistry. The molecular shape of PH3 affects its interactions with other atmospheric compounds, influencing its reactivity and lifetime in the atmosphere. Understanding these interactions can help researchers develop strategies to mitigate the environmental impact of PH3 and other phosphorous-containing compounds.
Future Directions in PH3 Research
The study of the molecular shape of PH3 is an active area of research with many potential directions. Future research can focus on understanding the detailed mechanisms of PH3 reactions, developing new applications, and exploring its environmental impact. Advances in computational chemistry and experimental techniques can provide new insights into the molecular shape and properties of PH3, leading to the development of innovative technologies and solutions.
One potential direction is the development of new catalysts using PH3 as a precursor. The molecular shape of PH3 allows it to interact with other molecules in specific ways, leading to the formation of catalysts with unique properties. Researchers can explore the use of PH3 in the synthesis of heterogeneous and homogeneous catalysts for various chemical reactions, including oxidation, reduction, and polymerization.
Another direction is the study of PH3 in atmospheric chemistry. Understanding the molecular shape and properties of PH3 can help researchers predict its behavior in the atmosphere and develop strategies to mitigate its environmental impact. Future research can focus on the interactions of PH3 with other atmospheric compounds, its role in the formation of aerosols, and its impact on climate change.
Additionally, the development of new materials using PH3 as a precursor is a promising area of research. The molecular shape of PH3 allows it to interact with other molecules in specific ways, leading to the formation of materials with unique properties. Researchers can explore the use of PH3 in the synthesis of semiconductors, polymers, and other materials for various applications, including electronics, energy storage, and environmental remediation.
📝 Note: Future research on PH3 can lead to the development of new catalysts, materials, and environmental solutions, contributing to advancements in various fields.
In conclusion, the molecular shape of PH3 is a critical factor in its chemical behavior and interactions. Understanding the trigonal pyramidal shape of PH3, influenced by the lone pair on the phosphorus atom, provides insights into its reactivity, solubility, and applications in various fields. Comparing PH3 with other molecules highlights the role of lone pairs in determining molecular shape and properties. The molecular shape of PH3 has significant implications in chemistry, materials science, and environmental studies, paving the way for future research and innovations. As research continues, the molecular shape of PH3 will remain a key area of study, driving advancements in technology and sustainability.
Related Terms:
- lewis dot diagram for ph3
- ph3 electron pair geometry
- ph3 electron geometry and molecular
- ph3 3d shape
- electron dot structure for ph3
- molecular geometry for ph3