Understanding the Lewis Structure of CCl4 is fundamental for anyone studying chemistry, particularly those delving into organic chemistry and molecular structures. Carbon tetrachloride (CCl4) is a significant compound with a rich history in various industrial applications. This blog post will guide you through the process of drawing the Lewis Structure of CCl4, explaining its molecular geometry, and exploring its properties and applications.
What is Carbon Tetrachloride (CCl4)?
Carbon tetrachloride, commonly known as CCl4, is a chemical compound consisting of one carbon atom bonded to four chlorine atoms. It is a colorless, non-flammable liquid with a sweet smell. Historically, CCl4 has been used as a solvent in various industries, including dry cleaning and fire extinguishers. However, due to its environmental and health impacts, its use has significantly declined.
Drawing the Lewis Structure of CCl4
To draw the Lewis Structure of CCl4, follow these steps:
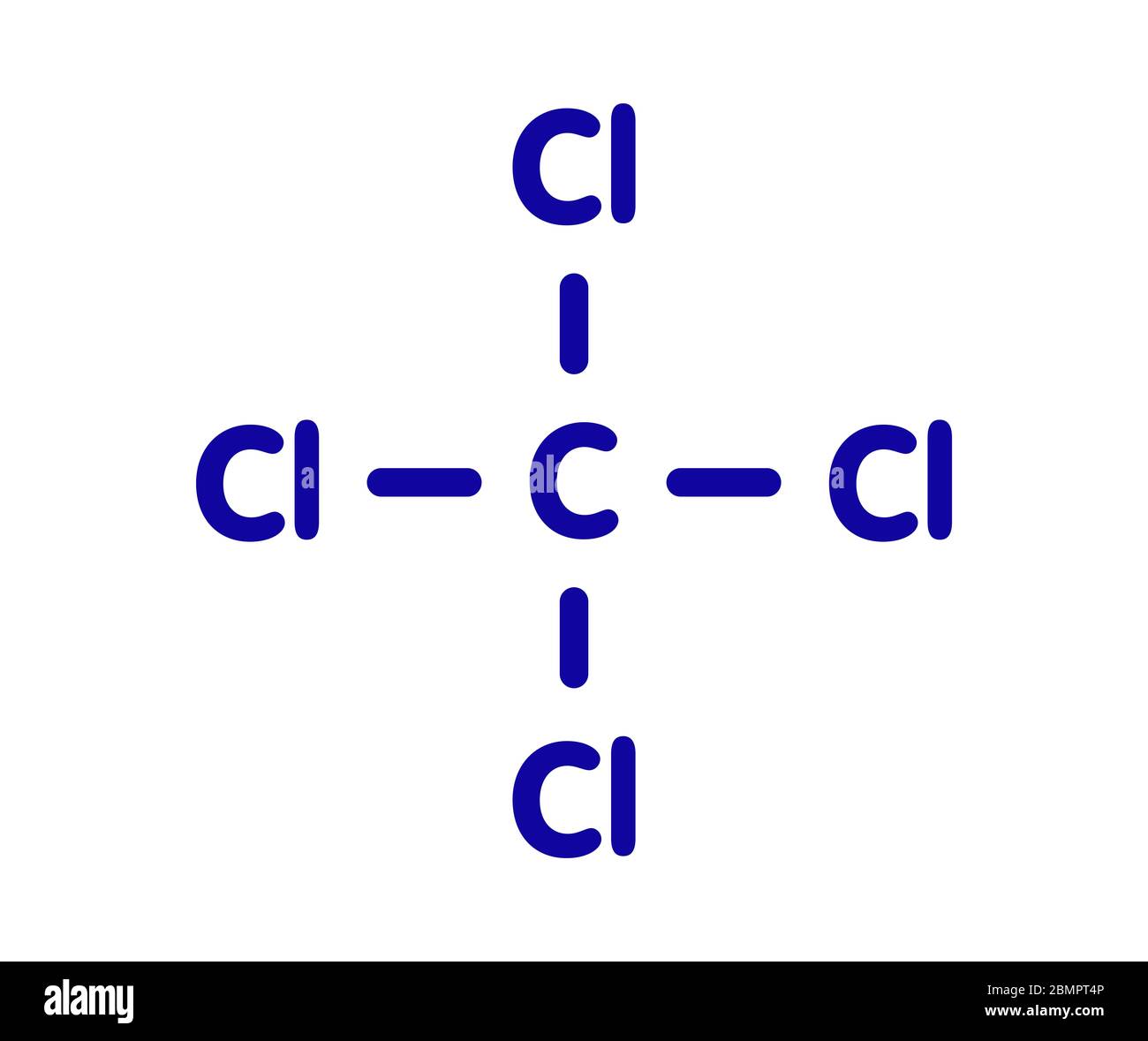
- Identify the central atom: In CCl4, the central atom is carbon (C).
- Count the total number of valence electrons: Carbon has 4 valence electrons, and each chlorine atom has 7 valence electrons. Therefore, the total number of valence electrons is 4 (from carbon) + 4 * 7 (from chlorine) = 32.
- Place the valence electrons around the central atom: Start by placing one pair of electrons between the carbon atom and each chlorine atom to form single bonds. This uses up 8 electrons (4 pairs).
- Distribute the remaining electrons: After forming the single bonds, you have 24 electrons left. These electrons will be placed as lone pairs on the chlorine atoms, giving each chlorine atom 3 lone pairs (6 electrons each).
Here is the Lewis Structure of CCl4:

Molecular Geometry of CCl4
The molecular geometry of CCl4 is tetrahedral. This means that the four chlorine atoms are arranged around the central carbon atom in a three-dimensional shape resembling a pyramid with a triangular base. The bond angles between the chlorine atoms are approximately 109.5 degrees, which is characteristic of a tetrahedral geometry.
This geometry is crucial for understanding the properties of CCl4, as it influences the molecule's polarity, reactivity, and other chemical behaviors.
Properties of CCl4
CCl4 exhibits several notable properties that make it a unique compound:
- Non-Polar Nature: Despite having polar C-Cl bonds, the symmetrical tetrahedral geometry of CCl4 results in a non-polar molecule. The dipole moments of the individual bonds cancel each other out, making the overall molecule non-polar.
- High Boiling Point: CCl4 has a relatively high boiling point of 76.72°C, which is higher than many other similar compounds. This is due to the strong London dispersion forces between the molecules.
- Density: CCl4 is denser than water, with a density of approximately 1.59 g/mL at room temperature.
- Solubility: CCl4 is insoluble in water but soluble in many organic solvents. This property makes it useful as a solvent in various chemical processes.
Applications of CCl4
Historically, CCl4 has been used in a variety of applications, although its use has declined due to environmental and health concerns:
- Solvent: CCl4 was widely used as a solvent in dry cleaning, degreasing, and other industrial processes. Its non-flammable nature made it a popular choice for these applications.
- Fire Extinguishers: CCl4 was used in fire extinguishers due to its ability to smother flames by displacing oxygen.
- Refrigerants: In the past, CCl4 was used as a refrigerant in cooling systems. However, its ozone-depleting properties led to its phase-out under the Montreal Protocol.
Environmental and Health Impacts
Despite its useful properties, CCl4 poses significant environmental and health risks:
- Ozone Depletion: CCl4 is a potent ozone-depleting substance. When released into the atmosphere, it breaks down and releases chlorine atoms that catalyze the destruction of ozone molecules.
- Health Hazards: Exposure to CCl4 can cause serious health issues, including liver and kidney damage, and it is classified as a carcinogen by the International Agency for Research on Cancer (IARC).
- Environmental Persistence: CCl4 is highly persistent in the environment, meaning it does not break down easily and can accumulate in soil and water, posing long-term risks to ecosystems.
📌 Note: Due to its environmental and health impacts, the use of CCl4 has been significantly restricted under international agreements such as the Montreal Protocol. Alternatives with lower environmental impact are now preferred.
Safety Precautions
Handling CCl4 requires strict safety precautions to minimize health and environmental risks:
- Personal Protective Equipment (PPE): Use appropriate PPE, including gloves, safety glasses, and lab coats, when handling CCl4.
- Ventilation: Ensure adequate ventilation to prevent the accumulation of CCl4 vapors in the workspace.
- Disposal: Dispose of CCl4 and its containers according to local regulations and guidelines to prevent environmental contamination.
- Emergency Procedures: Have emergency procedures in place in case of spills or accidental exposure, including access to safety showers and eyewash stations.
By following these precautions, the risks associated with handling CCl4 can be significantly reduced.
Alternatives to CCl4
Given the environmental and health concerns associated with CCl4, several alternatives have been developed:
- Hydrofluorocarbons (HFCs): HFCs are used as refrigerants and solvents, offering lower environmental impact compared to CCl4.
- Hydrochlorofluorocarbons (HCFCs): HCFCs are transitional replacements for CCl4, with lower ozone-depleting potential but still subject to phase-out under international agreements.
- Perfluorocarbons (PFCs): PFCs are used in various applications, including as solvents and refrigerants, and have lower environmental impact.
These alternatives provide safer and more environmentally friendly options for applications where CCl4 was previously used.
Conclusion
Understanding the Lewis Structure of CCl4 is essential for grasping its molecular properties and behaviors. The tetrahedral geometry of CCl4, along with its non-polar nature, contributes to its unique chemical characteristics. While CCl4 has historically been valuable in various industrial applications, its environmental and health impacts have led to a significant decline in its use. Alternatives with lower environmental impact are now preferred, ensuring safer and more sustainable practices. By adhering to safety precautions and exploring eco-friendly alternatives, we can mitigate the risks associated with CCl4 and promote a healthier environment.
Related Terms:
- clo lewis structure
- xef4 lewis structure
- c2h4 lewis structure
- ch3cl lewis structure
- ccl4 molecular geometry
- sicl4 lewis structure