In the realm of genetic testing, the Kras G12c Test has emerged as a pivotal tool for understanding and managing certain types of cancer. This test focuses on identifying specific mutations in the KRAS gene, particularly the G12C mutation, which is associated with various cancers, including lung and colorectal cancer. By detecting this mutation, healthcare providers can tailor treatment plans more effectively, improving patient outcomes.
Understanding the Kras G12c Mutation
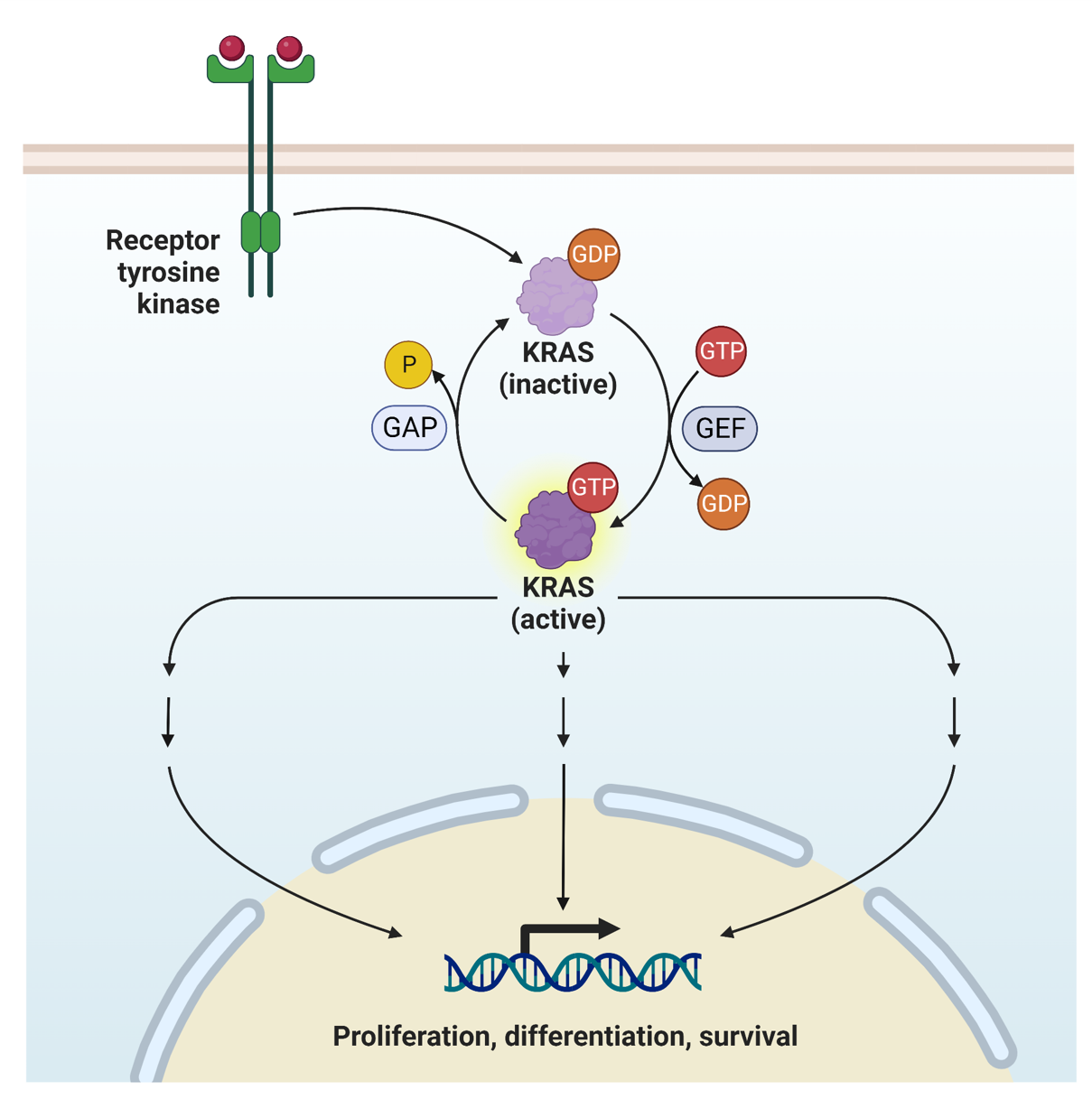
The KRAS gene plays a crucial role in cell signaling pathways that regulate cell growth and division. Mutations in this gene can lead to uncontrolled cell proliferation, a hallmark of cancer. The G12C mutation is one of the most common mutations in the KRAS gene, and it is particularly significant because it can be targeted by specific therapies.
When the Kras G12c Test is performed, it looks for the presence of this specific mutation in a patient's tumor tissue. The test involves analyzing a small sample of the tumor, which can be obtained through a biopsy. The sample is then subjected to molecular testing techniques, such as polymerase chain reaction (PCR) or next-generation sequencing (NGS), to detect the G12C mutation.
Importance of the Kras G12c Test in Cancer Treatment
The Kras G12c Test is not just a diagnostic tool; it is a critical component of personalized medicine. By identifying the G12C mutation, oncologists can determine the most effective treatment options for their patients. This is particularly important because traditional chemotherapy and radiation therapy may not be effective against tumors with this mutation. Instead, targeted therapies that specifically inhibit the mutated KRAS protein can be used.
One of the most notable targeted therapies for the G12C mutation is sotorasib, a drug that has shown promising results in clinical trials. Sotorasib works by binding to the mutated KRAS protein and preventing it from activating downstream signaling pathways that drive cancer growth. This targeted approach can lead to better outcomes for patients, including improved survival rates and reduced side effects compared to traditional treatments.
The Process of Kras G12c Testing
The process of performing a Kras G12c Test involves several steps, each crucial for accurate results. Here is an overview of the procedure:
- Sample Collection: A small sample of the tumor is collected through a biopsy. This can be done using various methods, depending on the location and type of tumor.
- Sample Preparation: The collected sample is prepared for molecular testing. This involves isolating the DNA from the tumor cells.
- Molecular Testing: The isolated DNA is analyzed using techniques such as PCR or NGS to detect the G12C mutation. These methods amplify the specific region of the KRAS gene and sequence it to identify any mutations.
- Result Interpretation: The results of the molecular testing are interpreted by a pathologist or geneticist. If the G12C mutation is detected, the patient may be eligible for targeted therapies.
📝 Note: The accuracy of the Kras G12c Test depends on the quality of the tumor sample and the sensitivity of the molecular testing methods used. It is essential to follow standardized protocols to ensure reliable results.
Clinical Implications of Kras G12c Testing
The clinical implications of the Kras G12c Test are far-reaching. For patients with the G12C mutation, the test provides a clear pathway to targeted therapies that can significantly improve their prognosis. However, it is important to note that not all patients with KRAS mutations will have the G12C mutation. Therefore, the test is a crucial step in determining the most appropriate treatment plan.
In addition to guiding treatment decisions, the Kras G12c Test can also inform clinical trials and research. By identifying patients with the G12C mutation, researchers can enroll them in studies aimed at developing new therapies or improving existing ones. This contributes to the broader understanding of KRAS mutations and their role in cancer.
Challenges and Limitations
While the Kras G12c Test offers significant benefits, it also comes with challenges and limitations. One of the primary challenges is the availability of the test. Not all healthcare facilities have the capability to perform molecular testing, which can limit access to this important diagnostic tool. Additionally, the cost of the test and subsequent targeted therapies can be a barrier for some patients.
Another limitation is the potential for false-negative results. If the tumor sample is not representative of the entire tumor or if the molecular testing methods are not sensitive enough, the test may fail to detect the G12C mutation. This underscores the importance of using high-quality samples and advanced testing techniques.
Furthermore, the effectiveness of targeted therapies for the G12C mutation can vary among patients. While sotorasib has shown promising results, not all patients will respond to the treatment. Ongoing research is needed to identify biomarkers that can predict response to targeted therapies and to develop new treatments for patients who do not respond to existing options.
Future Directions in Kras G12c Testing
The field of Kras G12c Testing is rapidly evolving, with several exciting developments on the horizon. One area of focus is the development of liquid biopsies, which involve analyzing circulating tumor DNA (ctDNA) in the bloodstream. Liquid biopsies offer a non-invasive alternative to traditional tissue biopsies and can provide real-time information about the tumor's genetic makeup.
Another promising area is the integration of Kras G12c Testing with other molecular tests. By combining the results of multiple tests, healthcare providers can gain a more comprehensive understanding of the tumor's genetic profile and tailor treatment plans accordingly. This multi-omic approach can lead to more personalized and effective cancer treatments.
Additionally, advancements in molecular testing technologies are expected to improve the sensitivity and specificity of Kras G12c Testing. New techniques, such as digital PCR and single-cell sequencing, can provide more detailed and accurate information about the tumor's genetic makeup, enhancing the precision of treatment decisions.
Finally, the development of new targeted therapies for the G12C mutation is a critical area of research. While sotorasib is currently the most prominent targeted therapy, ongoing clinical trials are exploring other drugs that may offer additional benefits. These efforts aim to expand the treatment options available to patients with the G12C mutation and improve overall outcomes.
In conclusion, the Kras G12c Test is a groundbreaking tool in the fight against cancer. By identifying the G12C mutation in the KRAS gene, this test enables healthcare providers to tailor treatment plans more effectively, improving patient outcomes. While challenges and limitations exist, ongoing research and technological advancements are paving the way for more precise and personalized cancer treatments. As our understanding of KRAS mutations continues to grow, the Kras G12c Test will play an increasingly important role in the management of cancer, offering hope to patients and their families.
Related Terms:
- g12c mutation in kras
- kras g12c mutation positive
- kras g12c mutation
- kras g12c prevalence chart
- kras g12c mutation lung cancer
- kras g12c mutation rate