Ammonia is a compound that has sparked considerable debate among chemists and students alike regarding its classification as an acid or a base. The question "Is Ammonia A Base?" is a fundamental one in the study of chemistry, particularly in the context of acid-base theories. This blog post aims to delve into the properties of ammonia, its behavior in aqueous solutions, and its classification according to different acid-base theories.
Understanding Ammonia
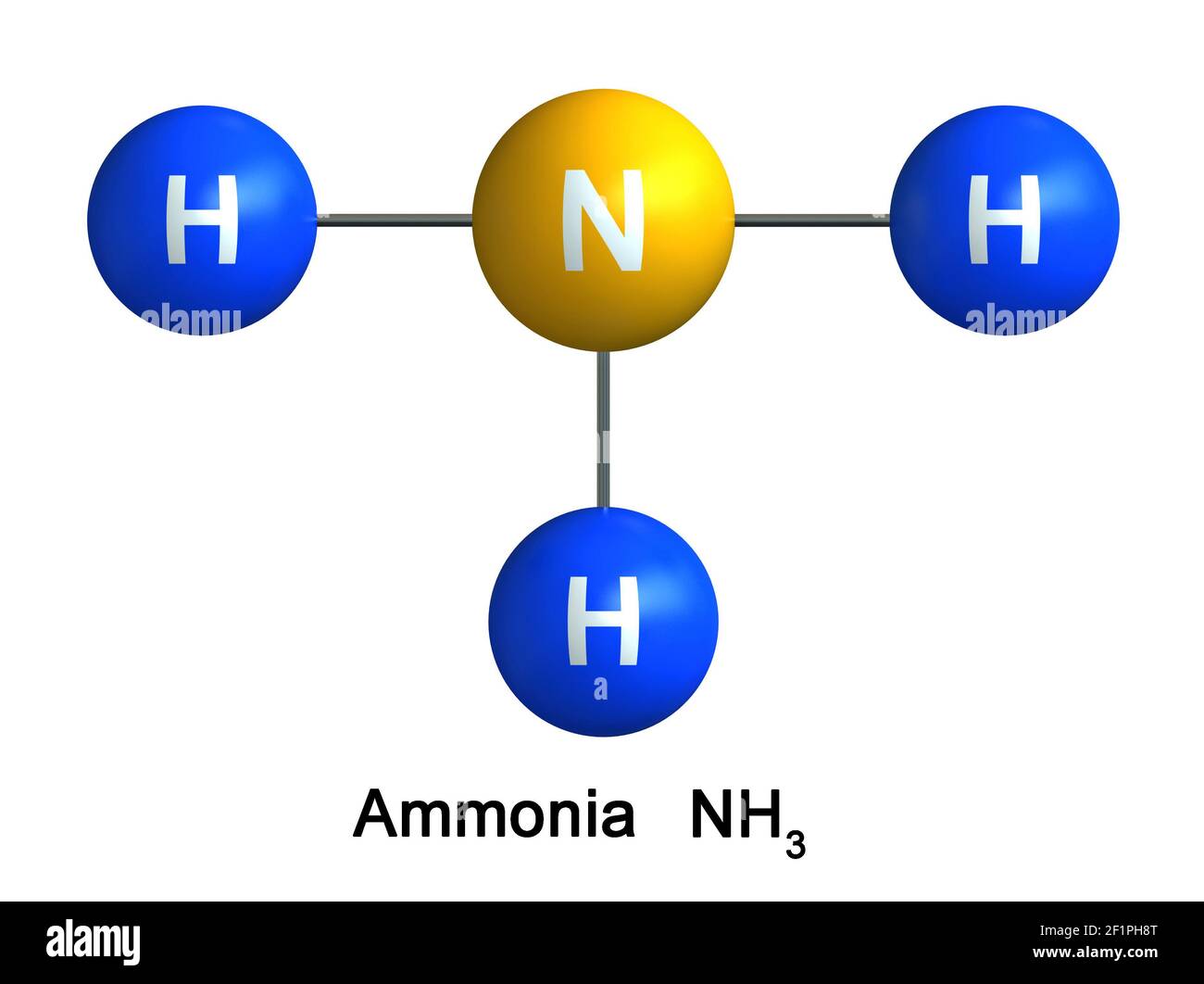
Ammonia, with the chemical formula NH3, is a colorless gas with a pungent odor. It is widely used in various industries, including agriculture, manufacturing, and cleaning products. In its pure form, ammonia is a gas, but it can be dissolved in water to form an aqueous solution known as ammonium hydroxide (NH4OH).
Acid-Base Theories
To understand whether ammonia is a base, it is essential to explore the different theories that define acids and bases. The three primary theories are:
- Arrhenius Theory: This theory defines acids as substances that produce hydrogen ions (H+) in aqueous solutions and bases as substances that produce hydroxide ions (OH-).
- Brønsted-Lowry Theory: This theory defines acids as proton (H+) donors and bases as proton acceptors.
- Lewis Theory: This theory defines acids as electron pair acceptors and bases as electron pair donors.
Ammonia as a Base According to the Arrhenius Theory
According to the Arrhenius theory, ammonia is not considered a base because it does not produce hydroxide ions (OH-) in aqueous solutions. Instead, it reacts with water to form ammonium ions (NH4+) and hydroxide ions:

NH3 + H2O ⇌ NH4+ + OH-
However, this reaction shows that ammonia can increase the concentration of hydroxide ions in a solution, which is a characteristic of a base.
Ammonia as a Base According to the Brønsted-Lowry Theory
According to the Brønsted-Lowry theory, ammonia is classified as a base because it accepts a proton (H+) from water to form ammonium ions (NH4+):
NH3 + H2O ⇌ NH4+ + OH-
In this reaction, ammonia acts as a proton acceptor, which aligns with the definition of a base in the Brønsted-Lowry theory.
Ammonia as a Base According to the Lewis Theory
According to the Lewis theory, ammonia is a base because it has a lone pair of electrons that it can donate to form a coordinate covalent bond. When ammonia reacts with a Lewis acid, such as boron trifluoride (BF3), it donates its lone pair of electrons to form a complex:
NH3 + BF3 → NH3BF3
In this reaction, ammonia acts as an electron pair donor, which is characteristic of a Lewis base.
Properties of Ammonia
To further understand why ammonia is considered a base, it is helpful to examine its properties:
- Basic Nature: Ammonia has a basic nature due to its ability to accept protons and donate electron pairs.
- Solubility: Ammonia is highly soluble in water, forming an aqueous solution that can conduct electricity due to the presence of ions.
- pH Value: An aqueous solution of ammonia has a pH greater than 7, indicating that it is basic.
- Reactivity: Ammonia reacts with acids to form ammonium salts, further confirming its basic nature.
Ammonia in Everyday Life
Ammonia is widely used in various applications due to its basic properties:
- Cleaning Products: Ammonia is a common ingredient in household cleaning products because of its ability to dissolve grease and grime.
- Agriculture: Ammonia is used as a fertilizer due to its high nitrogen content, which is essential for plant growth.
- Manufacturing: Ammonia is used in the production of various chemicals, including plastics, textiles, and explosives.
Safety Considerations
While ammonia has many useful applications, it is also important to handle it with care. Ammonia is a corrosive and irritating substance that can cause skin and eye irritation, as well as respiratory problems if inhaled. Proper safety measures, including the use of personal protective equipment (PPE) and adequate ventilation, should be taken when handling ammonia.
🛑 Note: Always store ammonia in a cool, well-ventilated area away from heat sources and incompatible substances.
Conclusion
In summary, ammonia is indeed a base according to the Brønsted-Lowry and Lewis theories. Its ability to accept protons and donate electron pairs makes it a versatile compound with numerous applications in industry and everyday life. Understanding the properties and behavior of ammonia is crucial for its safe and effective use. Whether you are a student studying chemistry or a professional working with ammonia, recognizing its basic nature is essential for a comprehensive understanding of this important compound.
Related Terms:
- is ammonia an alkali
- is sodium hydroxide a base
- is ammonia a strong base
- is ammonia a acid
- is ammonia basic or acidic
- is nh4 a base