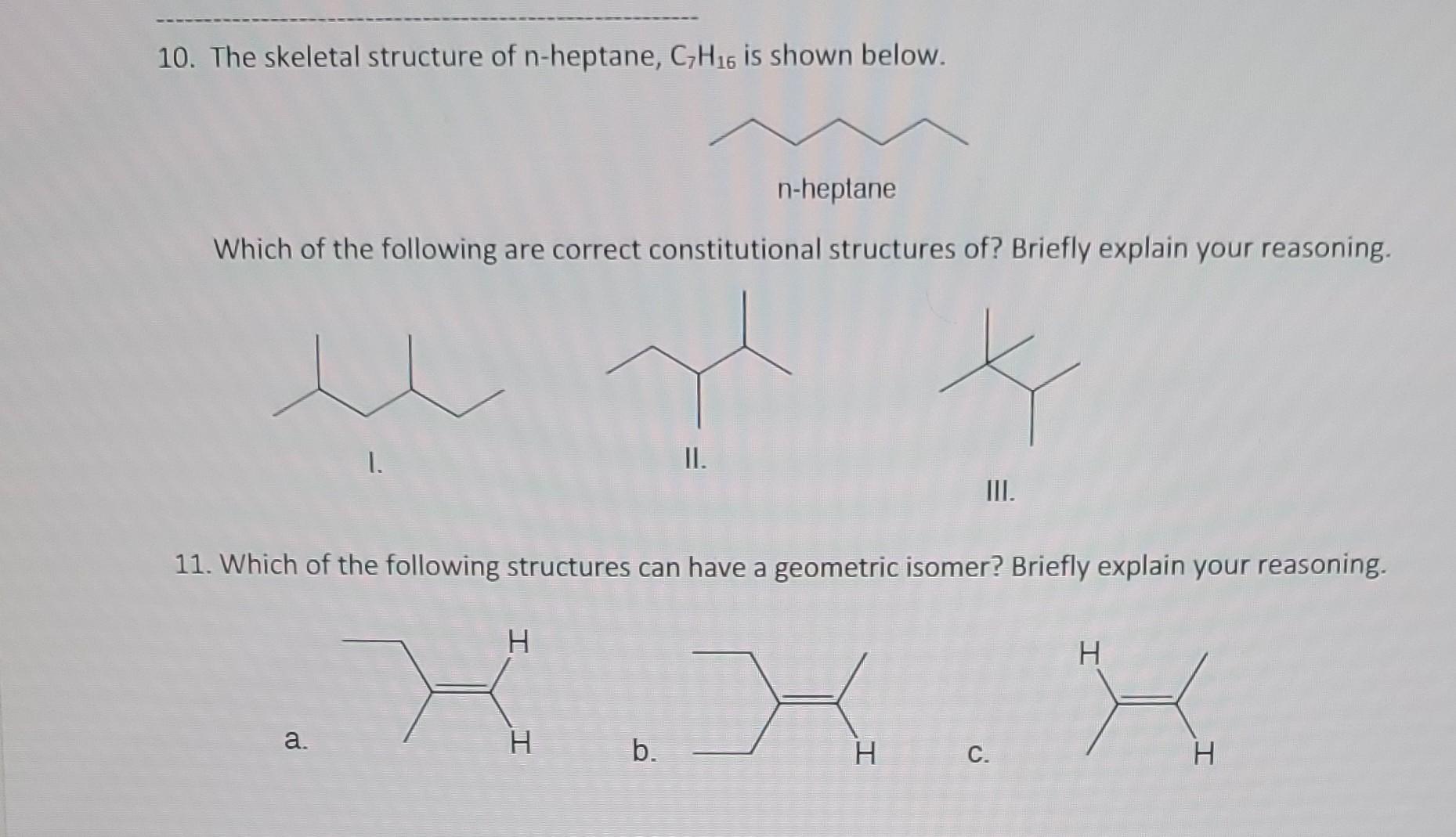
Heptane and n-heptane are both hydrocarbons with the chemical formula C7H16, but they differ in their molecular structure. Heptane is a straight-chain alkane, while n-heptane is a specific isomer of heptane with a linear structure. Understanding the differences and properties of these compounds is crucial in various industrial and scientific applications.
Understanding Heptane and N-Heptane
Heptane is a hydrocarbon that belongs to the alkane family. It is a saturated hydrocarbon, meaning it contains only single bonds between carbon atoms. Heptane can exist in several isomers, each with a different molecular structure but the same chemical formula. The most common isomers of heptane include:
- n-Heptane
- 2-Methylhexane
- 3-Methylhexane
- 2,2-Dimethylpentane
- 2,3-Dimethylpentane
- 2,4-Dimethylpentane
- 3,3-Dimethylpentane
- 3-Ethylpentane
Among these isomers, n-heptane is the straight-chain isomer, which means all carbon atoms are arranged in a linear fashion. This linear structure gives n-heptane unique properties that distinguish it from other heptane isomers.
Properties of Heptane and N-Heptane
Heptane and n-heptane share many properties due to their identical chemical formulas, but their structural differences lead to variations in certain characteristics. Here are some key properties:
| Property | Heptane | N-Heptane |
|---|---|---|
| Molecular Formula | C7H16 | C7H16 |
| Molecular Weight | 100.20 g/mol | 100.20 g/mol |
| Boiling Point | 98.4°C (209.1°F) | 98.4°C (209.1°F) |
| Melting Point | -90.6°C (-131.1°F) | -90.6°C (-131.1°F) |
| Density | 0.684 g/mL at 20°C | 0.684 g/mL at 20°C |
| Structure | Various isomers | Linear structure |
While the physical properties of heptane and n-heptane are similar, their chemical behaviors can differ due to their structural variations. For instance, n-heptane's linear structure makes it more susceptible to certain chemical reactions compared to branched isomers.
Applications of Heptane and N-Heptane
Heptane and n-heptane are used in various industries due to their unique properties. Some of the key applications include:
- Solvents: Both heptane and n-heptane are used as solvents in various industrial processes. Their ability to dissolve non-polar substances makes them ideal for cleaning and degreasing applications.
- Fuel Additives: Heptane is often used as a component in gasoline and other fuels. Its high octane rating makes it a valuable additive for improving fuel performance.
- Laboratory Reagents: N-heptane is commonly used in laboratory settings as a reference standard for octane rating. Its linear structure provides a consistent baseline for comparing the performance of different fuels.
- Chemical Synthesis: Heptane and n-heptane are used as starting materials in the synthesis of various chemicals. Their reactivity and stability make them suitable for a wide range of chemical reactions.
In addition to these applications, heptane and n-heptane are also used in the production of plastics, rubber, and other polymers. Their ability to act as a solvent and a reactant makes them versatile in the chemical industry.
Safety and Handling of Heptane and N-Heptane
Handling heptane and n-heptane requires careful attention to safety protocols. Both compounds are highly flammable and can pose significant risks if not handled properly. Here are some key safety considerations:
- Flammability: Heptane and n-heptane are highly flammable and can ignite easily. They should be stored in a cool, well-ventilated area away from sources of ignition.
- Ventilation: Proper ventilation is essential when handling heptane and n-heptane. Inhalation of vapors can cause dizziness, drowsiness, and other health issues.
- Personal Protective Equipment (PPE): Use appropriate PPE, including gloves, safety glasses, and protective clothing, when handling these compounds.
- Spill Management: In case of a spill, contain the area and use absorbent materials to clean up the liquid. Dispose of contaminated materials according to local regulations.
It is crucial to follow all safety guidelines and regulations when handling heptane and n-heptane to minimize risks and ensure safe working conditions.
🛑 Note: Always refer to the Material Safety Data Sheet (MSDS) for specific safety information and handling procedures.
Environmental Impact of Heptane and N-Heptane
Heptane and n-heptane, like many hydrocarbons, can have environmental impacts if not managed properly. Their use and disposal can contribute to air and water pollution. Here are some environmental considerations:
- Air Pollution: The combustion of heptane and n-heptane releases carbon dioxide, carbon monoxide, and other pollutants into the atmosphere. Proper emission controls are necessary to mitigate these effects.
- Water Pollution: Spills and improper disposal of heptane and n-heptane can contaminate water sources. These compounds are not easily biodegradable and can persist in the environment, posing risks to aquatic life.
- Soil Contamination: Heptane and n-heptane can seep into the soil, contaminating it and affecting plant growth. Proper containment and disposal methods are essential to prevent soil contamination.
To minimize the environmental impact, it is important to follow best practices for handling, storing, and disposing of heptane and n-heptane. Implementing proper waste management strategies and using emission control technologies can help reduce their environmental footprint.
🌿 Note: Always comply with local environmental regulations and guidelines for the safe disposal of heptane and n-heptane.
Future Trends in Heptane and N-Heptane Research
Research on heptane and n-heptane continues to evolve, driven by the need for more efficient and sustainable chemical processes. Some of the emerging trends in this field include:
- Green Chemistry: There is a growing focus on developing greener alternatives to traditional heptane and n-heptane-based processes. Researchers are exploring biodegradable solvents and more environmentally friendly synthesis methods.
- Catalytic Reactions: Advances in catalysis are enabling more efficient and selective reactions involving heptane and n-heptane. New catalysts are being developed to improve yield and reduce waste.
- Nanotechnology: Nanomaterials are being investigated for their potential to enhance the properties of heptane and n-heptane. Nanoparticles can act as catalysts or stabilizers, improving the performance of these compounds in various applications.
These trends highlight the ongoing efforts to improve the sustainability and efficiency of heptane and n-heptane-based processes. As research continues, we can expect to see more innovative applications and technologies emerge in this field.
Heptane and n-heptane are essential compounds in the chemical industry, with a wide range of applications and unique properties. Understanding their differences, properties, and safety considerations is crucial for their effective use. As research continues to advance, we can expect to see new and innovative applications of these compounds, contributing to various industrial and scientific fields.
Related Terms:
- n heptane vs n heptane
- heptane vs n hexane
- n heptane side effects
- heptane wikipedia
- what is n heptane