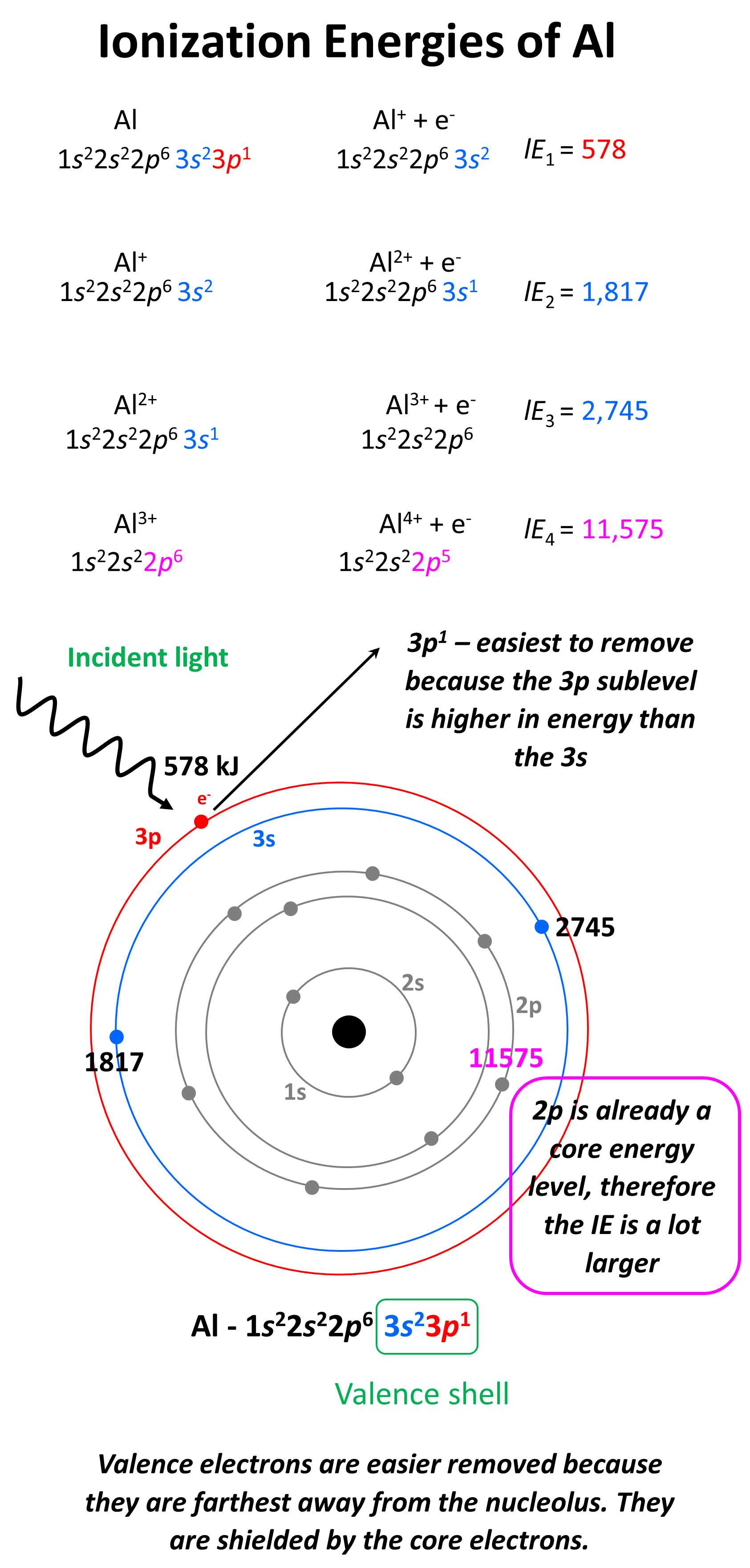
Understanding the First Ionization Potential Trend is crucial for anyone delving into the world of chemistry, particularly in the study of atomic and molecular structures. The first ionization potential refers to the energy required to remove the most loosely bound electron from a neutral atom or molecule in its ground state. This concept is fundamental in predicting the chemical behavior of elements and understanding the periodic trends in the periodic table.
What is First Ionization Potential?
The first ionization potential (IP) is a measure of the energy needed to remove an electron from an atom or molecule, converting it into a positively charged ion. It is typically expressed in electron volts (eV) or kilojoules per mole (kJ/mol). The process can be represented by the following equation:
X(g) → X⁺(g) + e⁻
Where X represents the atom or molecule, X⁺ is the resulting ion, and e⁻ is the electron removed.
Factors Affecting First Ionization Potential
Several factors influence the first ionization potential of an element. Understanding these factors helps in predicting the First Ionization Potential Trend across the periodic table.
- Atomic Radius: As the atomic radius increases, the distance between the nucleus and the outermost electron also increases. This results in a weaker attraction between the nucleus and the electron, making it easier to remove the electron and thus lowering the ionization potential.
- Nuclear Charge: The nuclear charge, or the number of protons in the nucleus, affects the ionization potential. A higher nuclear charge results in a stronger attraction between the nucleus and the electrons, making it harder to remove an electron and thus increasing the ionization potential.
- Electron Shielding: Electrons in inner shells can shield the outermost electrons from the nuclear charge, reducing the effective nuclear charge experienced by the outermost electron. This shielding effect can lower the ionization potential.
- Electron Configuration: The arrangement of electrons in different orbitals also plays a role. For example, elements with half-filled or fully filled orbitals tend to have higher ionization potentials due to the stability of these configurations.
The Periodic Trend of First Ionization Potential
The First Ionization Potential Trend across the periodic table follows a general pattern that can be explained by the factors mentioned above.
Across a Period
As you move from left to right across a period in the periodic table, the first ionization potential generally increases. This is due to the increasing nuclear charge and the decreasing atomic radius, which result in a stronger attraction between the nucleus and the outermost electron. For example, consider the elements in the second period:
| Element | First Ionization Potential (kJ/mol) |
|---|---|
| Lithium (Li) | 520 |
| Beryllium (Be) | 899 |
| Boron (B) | 801 |
| Carbon © | 1086 |
| Nitrogen (N) | 1402 |
| Oxygen (O) | 1314 |
| Fluorine (F) | 1681 |
| Neon (Ne) | 2081 |
Notice the general increase in ionization potential from lithium to neon, with some fluctuations due to electron configuration.
Down a Group
As you move down a group in the periodic table, the first ionization potential generally decreases. This is due to the increasing atomic radius and the shielding effect of inner electrons, which reduce the effective nuclear charge experienced by the outermost electron. For example, consider the elements in Group 1 (alkali metals):
| Element | First Ionization Potential (kJ/mol) |
|---|---|
| Lithium (Li) | 520 |
| Sodium (Na) | 496 |
| Potassium (K) | 419 |
| Rubidium (Rb) | 403 |
| Cesium (Cs) | 376 |
Notice the general decrease in ionization potential from lithium to cesium.
Exceptions to the Trend
While the general trends are useful for predicting ionization potentials, there are exceptions due to unique electron configurations. Some notable exceptions include:
- Beryllium and Boron: Beryllium has a higher ionization potential than boron, despite boron being to the right of beryllium in the periodic table. This is due to the stability of the fully filled 2s orbital in beryllium.
- Nitrogen and Oxygen: Nitrogen has a higher ionization potential than oxygen, despite oxygen being to the right of nitrogen. This is due to the half-filled 2p orbital in nitrogen, which provides extra stability.
- Group 2 Elements: Elements in Group 2 (alkaline earth metals) have higher ionization potentials than expected due to the fully filled s orbital.
📝 Note: These exceptions highlight the importance of electron configuration in determining ionization potentials.
Applications of First Ionization Potential
The concept of first ionization potential has numerous applications in chemistry and related fields. Some key applications include:
- Chemical Reactivity: Elements with low ionization potentials are more likely to lose electrons and form positive ions, making them good reducing agents. Conversely, elements with high ionization potentials are less likely to lose electrons and are good oxidizing agents.
- Periodic Table Trends: Understanding the First Ionization Potential Trend helps in predicting other periodic trends, such as atomic radius, electronegativity, and electron affinity.
- Spectroscopy: Ionization potentials are used in spectroscopy to identify elements based on their emission and absorption spectra.
- Material Science: In material science, ionization potentials are crucial for understanding the electronic properties of materials, which is essential for developing new materials with desired properties.
In the realm of chemistry, the First Ionization Potential Trend serves as a cornerstone for comprehending the behavior of elements and their compounds. By examining the factors that influence ionization potentials and understanding the periodic trends, chemists can make informed predictions about the reactivity and properties of elements. This knowledge is invaluable in various fields, from materials science to spectroscopy, and plays a crucial role in advancing our understanding of the chemical world.
In conclusion, the First Ionization Potential Trend is a fundamental concept in chemistry that provides insights into the behavior of elements. By understanding the factors that affect ionization potentials and the periodic trends, we can predict the chemical reactivity and properties of elements. This knowledge is essential for various applications, from materials science to spectroscopy, and continues to be a vital area of study in chemistry.
Related Terms:
- does ionization increase across period
- ionization potential in periodic table
- when does ionization energy increase
- ionization increasing on periodic table
- 3rd period ionization energy trend
- ionization trend on periodic table