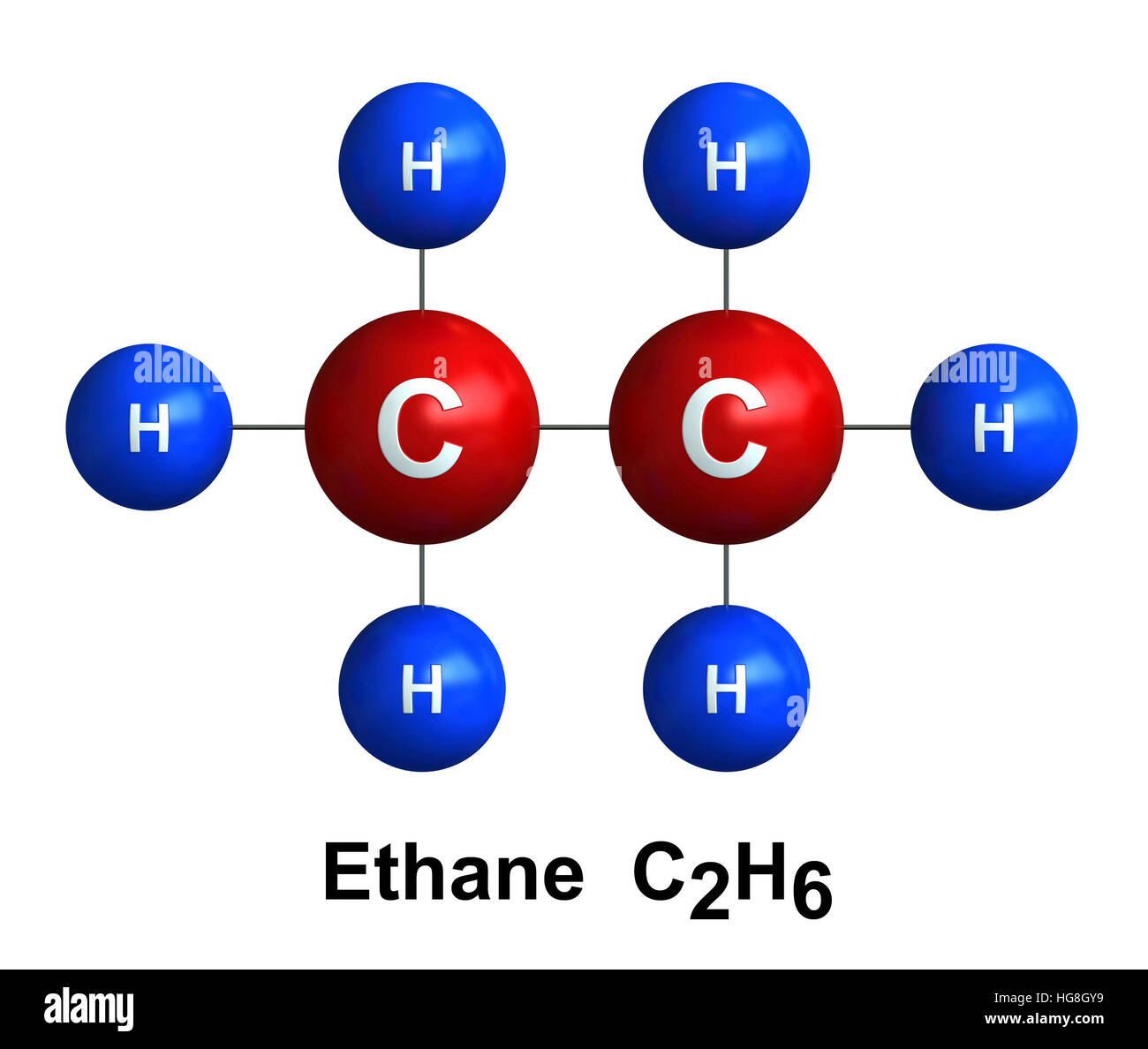
Understanding the ethane molar mass is fundamental in the field of chemistry, particularly when dealing with hydrocarbons and their properties. Ethane, a simple hydrocarbon with the chemical formula C2H6, is a colorless, odorless gas that is widely used in various industrial applications. This post delves into the significance of ethane molar mass, its calculation, and its applications in chemistry and industry.
What is Ethane?
Ethane is the second simplest hydrocarbon after methane. It consists of two carbon atoms bonded together, with each carbon atom also bonded to three hydrogen atoms. This structure gives ethane its chemical formula, C2H6. Ethane is a saturated hydrocarbon, meaning that all its carbon atoms are bonded to the maximum number of hydrogen atoms, forming single bonds.
Calculating the Ethane Molar Mass
The ethane molar mass is determined by summing the atomic masses of all the atoms in a molecule of ethane. The atomic mass of carbon © is approximately 12.01 g/mol, and the atomic mass of hydrogen (H) is approximately 1.008 g/mol. Ethane has two carbon atoms and six hydrogen atoms.
To calculate the ethane molar mass, use the following formula:
📝 Note: The atomic masses used here are standard values and may vary slightly depending on the source.
Molar mass of ethane = (2 × atomic mass of carbon) + (6 × atomic mass of hydrogen)
Molar mass of ethane = (2 × 12.01 g/mol) + (6 × 1.008 g/mol)
Molar mass of ethane = 24.02 g/mol + 6.048 g/mol
Molar mass of ethane = 30.068 g/mol
Therefore, the ethane molar mass is approximately 30.068 g/mol.
Importance of Ethane Molar Mass in Chemistry
The ethane molar mass is crucial in various chemical calculations and applications. Here are some key areas where the ethane molar mass plays a significant role:
- Stoichiometry: In chemical reactions involving ethane, the ethane molar mass is used to determine the amounts of reactants and products. This is essential for balancing chemical equations and predicting the outcomes of reactions.
- Gas Laws: The ethane molar mass is used in gas law calculations, such as those involving the ideal gas law (PV = nRT). Knowing the molar mass helps in determining the number of moles of ethane, which is necessary for these calculations.
- Thermodynamics: In thermodynamic calculations, the ethane molar mass is used to determine the enthalpy, entropy, and free energy changes in reactions involving ethane. These calculations are crucial for understanding the energy changes in chemical processes.
Applications of Ethane in Industry
Ethane is a valuable hydrocarbon with numerous industrial applications. Some of the key uses of ethane include:
- Petrochemical Industry: Ethane is a primary feedstock in the petrochemical industry. It is used to produce ethylene, which is a key building block for various plastics, such as polyethylene. The production of ethylene from ethane involves a process called steam cracking, where ethane is heated to high temperatures in the presence of steam to break down the molecules.
- Fuel: Ethane is used as a fuel in various applications, including heating and cooking. It is often mixed with other hydrocarbons, such as propane, to create liquefied petroleum gas (LPG). Ethane's high energy content makes it an efficient fuel source.
- Refrigeration: Ethane is used as a refrigerant in some cooling systems. Its low boiling point and non-toxic nature make it suitable for use in refrigeration and air conditioning systems.
Ethane Properties
Understanding the properties of ethane is essential for its effective use in various applications. Some of the key properties of ethane include:
| Property | Value |
|---|---|
| Molecular Formula | C2H6 |
| Molar Mass | 30.068 g/mol |
| Boiling Point | -88.6°C (-127.5°F) |
| Melting Point | -182.8°C (-297.0°F) |
| Density | 1.256 kg/m³ (at 0°C and 1 atm) |
| Solubility | Slightly soluble in water |
These properties make ethane a versatile hydrocarbon with a wide range of applications in industry and chemistry.
Safety Considerations
While ethane is a useful hydrocarbon, it is also important to consider safety when handling it. Ethane is a flammable gas and can form explosive mixtures with air. Proper safety measures should be taken to prevent accidents and ensure safe handling. Some safety considerations include:
- Ventilation: Ensure adequate ventilation when handling ethane to prevent the accumulation of flammable vapors.
- Storage: Store ethane in a cool, dry place away from sources of ignition. Use approved containers and follow safety guidelines for storage.
- Personal Protective Equipment (PPE): Use appropriate PPE, such as gloves and safety glasses, when handling ethane to protect against potential hazards.
By following these safety considerations, the risks associated with handling ethane can be minimized, ensuring safe and effective use.
Ethane is a versatile hydrocarbon with a wide range of applications in industry and chemistry. Understanding the ethane molar mass and its properties is essential for its effective use in various processes. From its role in the petrochemical industry to its use as a fuel and refrigerant, ethane plays a crucial role in modern society. By following safety guidelines and understanding its properties, ethane can be handled safely and effectively, contributing to its continued use in various applications.
Related Terms:
- o2 molar mass
- propylene molar mass
- hbr molar mass
- ethane density
- carbon dioxide molar mass
- butane molar mass