Understanding the difference between heterogeneous and homogeneous systems is crucial in various fields, including computer science, chemistry, and materials science. These terms describe the composition and structure of systems, and grasping their distinctions can provide deeper insights into how different materials and systems behave. This blog post will delve into the definitions, characteristics, and applications of heterogeneous and homogeneous systems, highlighting their unique properties and uses.
Understanding Homogeneous Systems
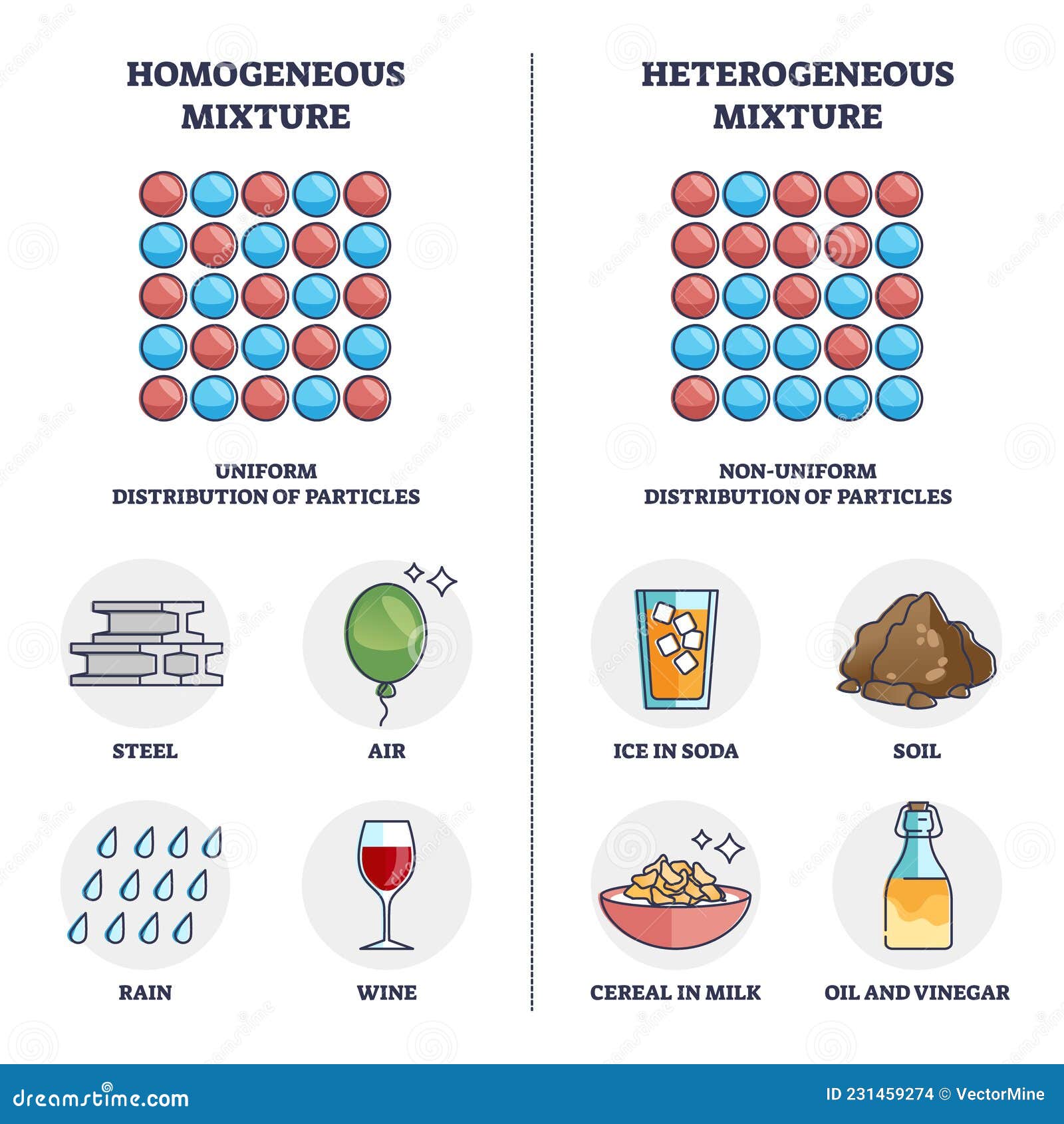
A homogeneous system is one in which the composition is uniform throughout. This means that any sample taken from the system will have the same properties and characteristics as any other sample. Homogeneous systems are often referred to as mixtures or solutions where the components are evenly distributed.
Examples of homogeneous systems include:
- Saltwater: The salt is evenly distributed throughout the water.
- Air: The gases (nitrogen, oxygen, etc.) are uniformly mixed.
- Alloys: Metals like brass (a mixture of copper and zinc) have a uniform composition.
Homogeneous systems are often easier to analyze and predict because their properties are consistent across the entire system. This uniformity makes them ideal for many scientific and industrial applications.
Characteristics of Homogeneous Systems
Homogeneous systems exhibit several key characteristics:
- Uniform Composition: The components are evenly distributed.
- Consistent Properties: Any sample from the system will have the same properties.
- Ease of Analysis: Predictable behavior makes them easier to study and use.
- Stability: The components do not separate over time.
These characteristics make homogeneous systems reliable for various applications, from pharmaceuticals to metallurgy.
Understanding Heterogeneous Systems
A heterogeneous system, on the other hand, is one in which the composition is not uniform. Different parts of the system can have different properties and characteristics. Heterogeneous systems are often referred to as mixtures where the components are not evenly distributed.
Examples of heterogeneous systems include:
- Rocks: Different minerals are distributed unevenly.
- Soil: Contains various particles like sand, clay, and organic matter.
- Granite: A mixture of different minerals like quartz, feldspar, and mica.
Heterogeneous systems are more complex to analyze due to their varied composition, but they offer unique properties that can be advantageous in certain applications.
Characteristics of Heterogeneous Systems
Heterogeneous systems have several distinct characteristics:
- Non-Uniform Composition: The components are not evenly distributed.
- Variable Properties: Different parts of the system can have different properties.
- Complexity: More challenging to analyze and predict.
- Potential for Separation: Components may separate over time.
These characteristics make heterogeneous systems suitable for applications where diversity in properties is beneficial, such as in construction materials and composite manufacturing.
Applications of Homogeneous and Heterogeneous Systems
The difference between heterogeneous and homogeneous systems is evident in their applications. Homogeneous systems are often used in scenarios where consistency and predictability are crucial. For example, in the pharmaceutical industry, homogeneous mixtures ensure that each dose of a medication has the same concentration of active ingredients. In metallurgy, homogeneous alloys provide consistent mechanical properties.
Heterogeneous systems, however, are used where varied properties are advantageous. In construction, heterogeneous materials like concrete (a mixture of cement, aggregates, and water) provide strength and durability. In environmental science, heterogeneous soil compositions support diverse ecosystems.
Comparing Homogeneous and Heterogeneous Systems
To better understand the difference between heterogeneous and homogeneous systems, let's compare them side by side:
| Characteristic | Homogeneous Systems | Heterogeneous Systems |
|---|---|---|
| Composition | Uniform | Non-Uniform |
| Properties | Consistent | Variable |
| Ease of Analysis | Easier | More Complex |
| Stability | Stable | May Separate Over Time |
| Examples | Saltwater, Air, Alloys | Rocks, Soil, Granite |
This comparison highlights the fundamental difference between heterogeneous and homogeneous systems, emphasizing their unique properties and applications.
💡 Note: The choice between using a homogeneous or heterogeneous system depends on the specific requirements of the application. Understanding the characteristics of each type can help in making informed decisions.
In the field of materials science, the difference between heterogeneous and homogeneous systems is particularly important. Homogeneous materials are often preferred for their consistent properties, making them ideal for applications where reliability is key. For example, in the aerospace industry, homogeneous materials like titanium alloys are used for their consistent strength and durability.
Heterogeneous materials, on the other hand, are valued for their diverse properties. In the automotive industry, composite materials made from a mixture of different fibers and resins are used to create lightweight and strong components. These materials offer a combination of properties that would be difficult to achieve with a single homogeneous material.
In chemistry, the difference between heterogeneous and homogeneous systems is crucial for understanding reactions and processes. Homogeneous reactions occur in a single phase, such as a solution, where the reactants are evenly distributed. This uniformity allows for predictable reaction rates and outcomes. For example, the reaction between hydrochloric acid and sodium hydroxide in water is a homogeneous reaction, resulting in the formation of sodium chloride and water.
Heterogeneous reactions, however, involve multiple phases, such as a solid and a liquid. These reactions can be more complex due to the varied properties of the different phases. For example, the reaction between calcium carbonate (a solid) and hydrochloric acid (a liquid) is a heterogeneous reaction, resulting in the formation of calcium chloride, water, and carbon dioxide. The reaction rate and outcome can be influenced by factors such as the surface area of the solid and the concentration of the acid.
In environmental science, the difference between heterogeneous and homogeneous systems is important for understanding natural processes and ecosystems. Homogeneous systems, such as the atmosphere, have consistent properties that can be modeled and predicted. This uniformity allows for accurate weather forecasting and climate modeling.
Heterogeneous systems, such as soil and water bodies, have varied properties that support diverse ecosystems. The composition of soil, for example, can influence plant growth and nutrient cycling. The varied properties of water bodies, such as lakes and rivers, support a wide range of aquatic life.
In conclusion, understanding the difference between heterogeneous and homogeneous systems is essential for various fields, from materials science to environmental science. Homogeneous systems offer consistency and predictability, making them ideal for applications where reliability is key. Heterogeneous systems, on the other hand, provide diverse properties that can be advantageous in certain applications. By recognizing the unique characteristics and applications of each type, we can make informed decisions and advance our understanding of the natural and engineered world.
Related Terms:
- homogeneous vs heterogeneous mixture chemistry
- difference between heterogeneous homogeneous examples
- heterogeneous vs homogeneous meaning
- heterogeneous vs homogeneous examples
- homogeneous vs heterogeneous products
- heterogeneous vs homogeneous simple