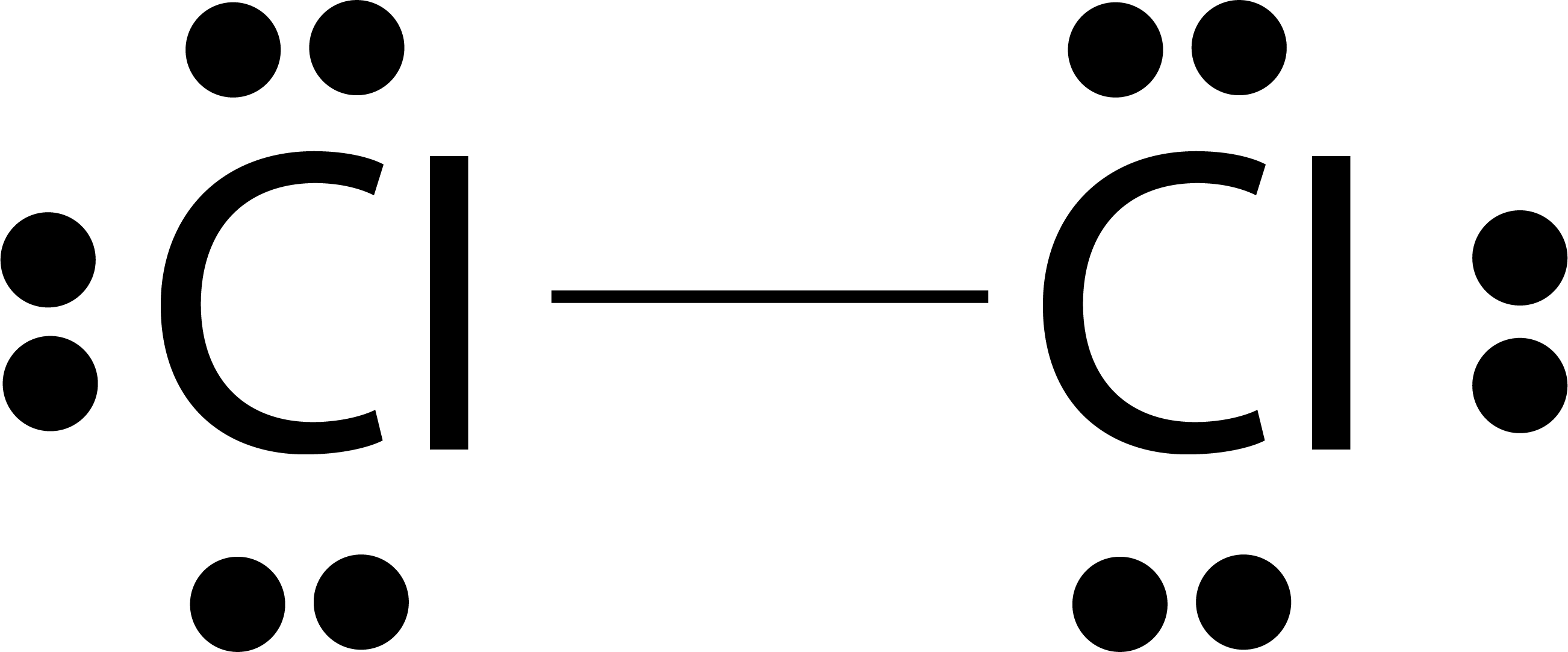Understanding the Cl2s Lewis Structure is fundamental for anyone studying chemistry, as it provides a visual representation of how atoms bond together to form molecules. This structure helps in predicting the properties and behavior of molecules, making it an essential tool for chemists and students alike. In this post, we will delve into the details of the Cl2s Lewis Structure, its significance, and how to draw it step by step.
What is the Lewis Structure?
The Lewis structure, also known as the Lewis dot structure, is a diagram that shows the bonding between atoms of a molecule and the lone pairs of electrons that may exist in the molecule. It was developed by Gilbert N. Lewis in 1916 and is a crucial concept in understanding chemical bonding. The structure helps in visualizing the arrangement of valence electrons around atoms, which is essential for predicting molecular geometry and reactivity.
Understanding the Cl2 Molecule
The Cl2 molecule, or dichlorine, is a diatomic molecule consisting of two chlorine atoms bonded together. Chlorine is a halogen element with the atomic number 17 and an electronic configuration of [Ne] 3s²3p⁵. This configuration indicates that chlorine has seven valence electrons, which are the electrons in the outermost shell and are involved in chemical bonding.
Drawing the Cl2s Lewis Structure
Drawing the Cl2s Lewis Structure involves several steps. Here’s a detailed guide to help you through the process:
Step 1: Determine the Total Number of Valence Electrons
Each chlorine atom has seven valence electrons. Since there are two chlorine atoms in the Cl2 molecule, the total number of valence electrons is:
7 (from the first Cl atom) + 7 (from the second Cl atom) = 14 valence electrons
Step 2: Choose the Central Atom
In the case of Cl2, there is no central atom because both chlorine atoms are identical and bonded directly to each other.
Step 3: Connect the Atoms with Single Bonds
Draw a single bond between the two chlorine atoms. A single bond consists of two electrons, so this step uses up 2 of the 14 valence electrons.
Step 4: Distribute the Remaining Valence Electrons
After placing the single bond, you have 12 valence electrons left to distribute. Place these electrons around the atoms to complete their octets (eight electrons in the outermost shell). Each chlorine atom will have three lone pairs of electrons (six electrons) in addition to the two electrons in the single bond.
Step 5: Verify the Octet Rule
The octet rule states that atoms tend to form bonds in such a way that they have eight electrons in their valence shell. For chlorine, this rule is satisfied as each atom has seven valence electrons plus one from the bond, making a total of eight.
The Final Cl2s Lewis Structure
The final Cl2s Lewis Structure will look like this:
Cl:Cl
Each chlorine atom has three lone pairs of electrons and one shared pair in the single bond.
Significance of the Cl2s Lewis Structure
The Cl2s Lewis Structure is significant for several reasons:
- Predicting Molecular Geometry: The Lewis structure helps in predicting the shape of the molecule. In the case of Cl2, the linear geometry is evident from the structure.
- Understanding Bonding: It provides insights into the type of bonding (single, double, or triple) and the number of bonds between atoms.
- Electronegativity and Polarity: The structure helps in understanding the electronegativity differences between atoms and predicting the polarity of the molecule.
- Reactivity: It aids in predicting the reactivity of the molecule by showing the availability of lone pairs and the stability of the bonds.
Common Mistakes to Avoid
When drawing the Cl2s Lewis Structure, it’s important to avoid common mistakes:
- Incorrect Valence Electrons: Ensure you count the correct number of valence electrons for each atom.
- Improper Bonding: Make sure to place the correct number of bonds between atoms.
- Ignoring Lone Pairs: Do not forget to distribute the remaining valence electrons as lone pairs around the atoms.
📝 Note: Always double-check your Lewis structure to ensure it adheres to the octet rule and correctly represents the bonding and lone pairs.
Examples of Other Lewis Structures
To further understand the concept, let’s look at a few more examples of Lewis structures:
H2O (Water)
Water has two hydrogen atoms and one oxygen atom. Oxygen has six valence electrons, and each hydrogen has one, making a total of eight valence electrons.
H-O-H
Oxygen has two lone pairs and two single bonds with hydrogen atoms.
CO2 (Carbon Dioxide)
Carbon dioxide has one carbon atom and two oxygen atoms. Carbon has four valence electrons, and each oxygen has six, making a total of 16 valence electrons.
O=C=O
Carbon forms double bonds with each oxygen atom, and each oxygen has two lone pairs.
NH3 (Ammonia)
Ammonia has one nitrogen atom and three hydrogen atoms. Nitrogen has five valence electrons, and each hydrogen has one, making a total of eight valence electrons.
H-N-H
H
Nitrogen has one lone pair and three single bonds with hydrogen atoms.
Conclusion
The Cl2s Lewis Structure is a fundamental concept in chemistry that helps in understanding the bonding and properties of molecules. By following the steps outlined above, you can accurately draw the Lewis structure for Cl2 and other molecules. This knowledge is crucial for predicting molecular geometry, reactivity, and other chemical properties. Mastering the Lewis structure is a key step in your journey to understanding chemistry.
Related Terms:
- cl2 lewis molecular structure
- cl2 molecule lewis structure
- lewis structure for chlorine molecule
- lewis diagram for cl2
- cl2 electron configuration
- cl2 dot cross diagram