Understanding the molecular geometry of a compound is crucial for predicting its chemical behavior and physical properties. One of the most fundamental concepts in this area is the CH4 molecular geometry, which refers to the three-dimensional arrangement of atoms in a methane molecule. This structure is a cornerstone in the study of chemistry, providing insights into bonding, reactivity, and the overall stability of molecules.
Understanding CH4 Molecular Geometry
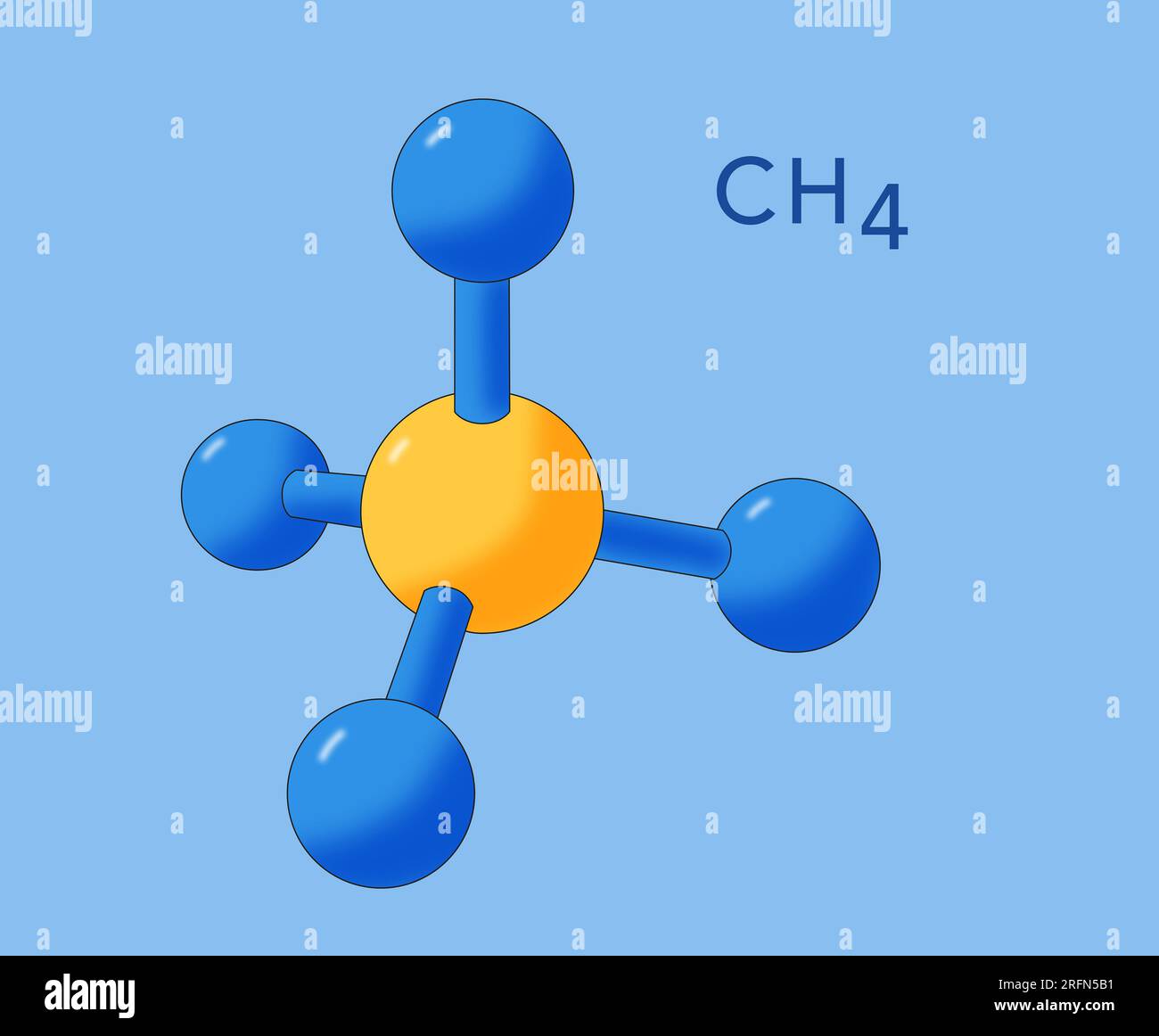
Methane (CH4) is the simplest hydrocarbon, consisting of one carbon atom bonded to four hydrogen atoms. The molecular geometry of CH4 is tetrahedral, meaning the four hydrogen atoms are positioned at the vertices of a tetrahedron, with the carbon atom at the center. This arrangement is a direct result of the valence shell electron pair repulsion (VSEPR) theory, which predicts that electron pairs around a central atom will repel each other and adopt a geometry that minimizes this repulsion.
The VSEPR Theory and CH4 Molecular Geometry
The VSEPR theory is a powerful tool for predicting the shapes of molecules based on the repulsion between electron pairs. In the case of CH4, the central carbon atom has four valence electrons, and each hydrogen atom contributes one electron to form a covalent bond. This results in four bonding pairs of electrons around the carbon atom. According to VSEPR theory, these four electron pairs will repel each other and arrange themselves in a tetrahedral geometry to minimize repulsion.
This tetrahedral arrangement ensures that the bond angles between the hydrogen atoms are approximately 109.5 degrees, which is the ideal angle for minimizing electron pair repulsion. The tetrahedral geometry of CH4 is stable and symmetrical, contributing to its non-polar nature and low reactivity.
Bonding in CH4
The bonding in CH4 is purely covalent, with each hydrogen atom sharing one electron with the carbon atom to form a single covalent bond. The carbon atom, with its four valence electrons, forms four single bonds with the hydrogen atoms. This results in a total of eight electrons in the valence shell of the carbon atom, achieving a stable octet configuration.
The covalent bonds in CH4 are strong and directional, contributing to the molecule's stability. The tetrahedral geometry ensures that the bonds are evenly distributed around the carbon atom, minimizing any potential strain or instability.
Properties of CH4
The CH4 molecular geometry significantly influences its physical and chemical properties. Some key properties of methane include:
- Non-polar Nature: Due to its symmetrical tetrahedral structure, methane is a non-polar molecule. This means it has no net dipole moment, making it insoluble in polar solvents like water.
- Low Reactivity: The strong covalent bonds and stable electron configuration make methane relatively inert under normal conditions. It does not readily react with other substances unless subjected to high temperatures or catalysts.
- Boiling and Melting Points: Methane has a very low boiling point (-161.5°C) and melting point (-182.5°C), reflecting its weak intermolecular forces. This makes it a gas at room temperature.
- Flammability: Methane is highly flammable and burns in the presence of oxygen to produce carbon dioxide and water. This property makes it a valuable fuel source.
Applications of CH4
Methane has numerous applications across various industries due to its unique properties. Some of the most significant applications include:
- Energy Source: Methane is a primary component of natural gas, which is widely used as a fuel for heating, cooking, and electricity generation. Its high energy content and clean-burning nature make it an attractive option for energy production.
- Chemical Industry: Methane is a key feedstock in the chemical industry, used in the production of various chemicals, including ammonia, methanol, and hydrogen. These chemicals are essential for manufacturing fertilizers, plastics, and other industrial products.
- Transportation Fuel: Compressed natural gas (CNG) and liquefied natural gas (LNG) are used as alternative fuels for vehicles, reducing emissions and dependence on petroleum-based fuels.
- Environmental Impact: While methane is a valuable resource, it is also a potent greenhouse gas. Understanding its molecular geometry and properties is crucial for developing strategies to mitigate its environmental impact, such as capturing and utilizing methane emissions from landfills and agricultural sources.
Comparing CH4 with Other Hydrocarbons
To better understand the significance of CH4 molecular geometry, it is helpful to compare methane with other hydrocarbons. The table below highlights some key differences:
| Hydrocarbon | Molecular Formula | Molecular Geometry | Bonding | Polarity |
|---|---|---|---|---|
| Methane | CH4 | Tetrahedral | Single covalent bonds | Non-polar |
| Ethane | C2H6 | Tetrahedral (around each carbon) | Single covalent bonds | Non-polar |
| Ethane | C2H4 | Trigonal planar (around each carbon) | One double bond and two single bonds | Non-polar |
| Propane | C3H8 | Tetrahedral (around each carbon) | Single covalent bonds | Non-polar |
As seen in the table, the molecular geometry of hydrocarbons varies with the number of carbon atoms and the type of bonding. Methane's tetrahedral geometry is unique and contributes to its distinct properties compared to other hydrocarbons.
📝 Note: The comparison table provides a quick reference for understanding how molecular geometry changes with different hydrocarbons. This can be useful for students and researchers studying the properties and behaviors of various hydrocarbons.
Experimental Techniques for Studying CH4 Molecular Geometry
Several experimental techniques are used to study the molecular geometry of CH4 and other compounds. These techniques provide valuable insights into the three-dimensional structure and bonding of molecules. Some of the most commonly used techniques include:
- X-ray Crystallography: This technique involves bombarding a crystal of the compound with X-rays and analyzing the diffraction pattern to determine the arrangement of atoms in the crystal lattice. While X-ray crystallography is not typically used for gases like methane, it is a powerful tool for studying the solid-state structures of other compounds.
- Electron Diffraction: Electron diffraction is used to study the structure of gases and liquids. In this technique, a beam of electrons is directed at the sample, and the diffraction pattern is analyzed to determine the molecular geometry.
- Microwave Spectroscopy: This technique involves measuring the absorption of microwave radiation by the molecule. The rotational energy levels of the molecule provide information about its geometry and bonding.
- Infrared Spectroscopy: Infrared spectroscopy measures the absorption of infrared radiation by the molecule, providing information about the vibrational modes and bonding within the molecule.
These experimental techniques, along with theoretical calculations, provide a comprehensive understanding of the CH4 molecular geometry and its implications for chemical behavior and physical properties.
📝 Note: Experimental techniques are essential for validating theoretical predictions about molecular geometry. They provide empirical data that can be used to refine and improve theoretical models.
Theoretical Calculations and Modeling
In addition to experimental techniques, theoretical calculations and modeling play a crucial role in studying the molecular geometry of CH4. Computational chemistry methods, such as density functional theory (DFT) and ab initio calculations, are used to predict the structure, energy, and properties of molecules. These methods involve solving the Schrödinger equation for the molecule to determine its electronic structure and geometry.
Theoretical calculations provide valuable insights into the bonding and electronic structure of CH4, complementing experimental data. They allow researchers to explore the effects of different conditions, such as temperature and pressure, on the molecular geometry and properties of methane.
One of the key advantages of theoretical calculations is their ability to predict the behavior of molecules under conditions that are difficult or impossible to achieve experimentally. For example, calculations can be used to study the behavior of methane at extremely high pressures or temperatures, providing insights into its potential applications in extreme environments.
Moreover, theoretical calculations can be used to design new materials and compounds with specific properties. By understanding the molecular geometry and bonding of CH4, researchers can develop new materials with enhanced stability, reactivity, or other desirable properties.
📝 Note: Theoretical calculations are a powerful tool for studying molecular geometry and properties. They complement experimental techniques and provide insights that are difficult or impossible to obtain through experimentation alone.
In conclusion, the CH4 molecular geometry is a fundamental concept in chemistry that provides insights into the bonding, reactivity, and stability of methane. Understanding this geometry is crucial for predicting the chemical behavior and physical properties of methane, as well as for developing new applications and technologies. The VSEPR theory, experimental techniques, and theoretical calculations all contribute to our understanding of the molecular geometry of CH4, highlighting its importance in the study of chemistry.
Related Terms:
- 3d molecular structure of ch4
- electron domain geometry of ch4
- ch4 lewis structure diagram
- ch4 molecular geometry shape
- molecular structure of ch4
- electron geometry for ch4