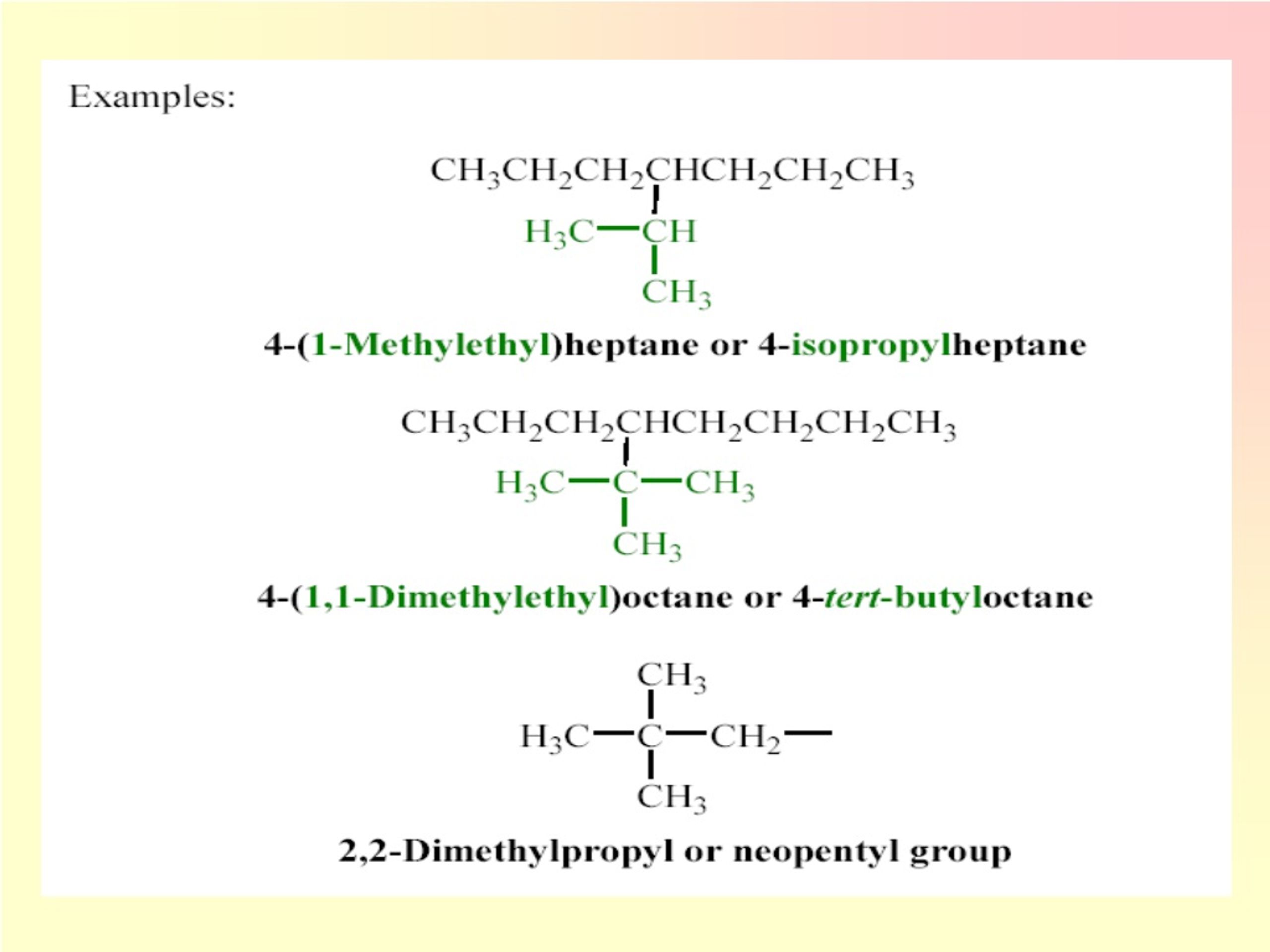
Understanding the Carbon Group Number is crucial for anyone involved in chemistry, particularly in organic chemistry. The Carbon Group Number refers to the number of carbon atoms in a molecule or compound. This number is fundamental in determining the properties, behavior, and potential applications of various organic compounds. Whether you are a student, researcher, or professional in the field, grasping the concept of the Carbon Group Number can significantly enhance your understanding of organic chemistry.
What is the Carbon Group Number?
The Carbon Group Number is a simple yet powerful concept that helps chemists classify and study organic compounds. It represents the total number of carbon atoms present in a molecule. This number is essential for several reasons:
- It aids in identifying the molecular formula of a compound.
- It helps in predicting the physical and chemical properties of the compound.
- It is crucial in understanding the reactivity and stability of organic molecules.
Importance of the Carbon Group Number in Organic Chemistry
The Carbon Group Number plays a pivotal role in various aspects of organic chemistry. Here are some key areas where it is particularly important:
Molecular Structure and Formula
The Carbon Group Number is directly related to the molecular formula of a compound. For example, methane (CH₄) has a Carbon Group Number of 1, while ethane (C₂H₆) has a Carbon Group Number of 2. Knowing the Carbon Group Number helps in writing the correct molecular formula, which is the foundation for further chemical analysis.
Physical Properties
The physical properties of organic compounds, such as boiling point, melting point, and solubility, are often correlated with the Carbon Group Number. Generally, as the Carbon Group Number increases, the boiling point and melting point of the compound also increase. This is due to the increased intermolecular forces, such as van der Waals forces, which become stronger with longer carbon chains.
Chemical Reactivity
The Carbon Group Number also influences the chemical reactivity of organic compounds. For instance, shorter carbon chains (lower Carbon Group Number) tend to be more reactive due to the higher strain and reactivity of the carbon-carbon bonds. Longer carbon chains (higher Carbon Group Number) may exhibit different reactivity patterns, often requiring more energy to break the bonds.
Biological and Industrial Applications
In biological and industrial settings, the Carbon Group Number is crucial for understanding the behavior and applications of organic compounds. For example, in the pharmaceutical industry, the Carbon Group Number helps in designing drugs with specific properties. In the petroleum industry, it aids in refining processes and understanding the composition of various hydrocarbons.
Determining the Carbon Group Number
Determining the Carbon Group Number of a compound involves identifying the number of carbon atoms in its molecular structure. This can be done through various methods, including:
Molecular Formula Analysis
One of the simplest ways to determine the Carbon Group Number is by analyzing the molecular formula. The number of carbon atoms in the formula directly gives the Carbon Group Number. For example, in the molecular formula C₆H₁₂O₆ (glucose), the Carbon Group Number is 6.
Spectroscopic Techniques
Spectroscopic techniques, such as Nuclear Magnetic Resonance (NMR) and Infrared (IR) spectroscopy, can provide detailed information about the molecular structure, including the number of carbon atoms. These techniques are particularly useful for complex molecules where the molecular formula alone may not be sufficient.
Mass Spectrometry
Mass spectrometry is another powerful tool for determining the Carbon Group Number. By analyzing the mass-to-charge ratio of the molecular ions, chemists can deduce the number of carbon atoms in the molecule. This method is highly accurate and can be used for a wide range of compounds.
Examples of Compounds with Different Carbon Group Numbers
To better understand the concept of the Carbon Group Number, let’s look at some examples of compounds with different Carbon Group Numbers:
| Compound | Molecular Formula | Carbon Group Number |
|---|---|---|
| Methane | CH₄ | 1 |
| Ethane | C₂H₆ | 2 |
| Propane | C₃H₈ | 3 |
| Butane | C₄H₁₀ | 4 |
| Pentane | C₅H₁₂ | 5 |
| Hexane | C₆H₁₄ | 6 |
📝 Note: The table above illustrates simple hydrocarbons, but the concept of the Carbon Group Number applies to all organic compounds, including those with functional groups and heteroatoms.
Applications of the Carbon Group Number
The Carbon Group Number has wide-ranging applications in various fields. Here are some key areas where it is particularly useful:
Pharmaceutical Industry
In the pharmaceutical industry, the Carbon Group Number is crucial for drug design and development. Understanding the Carbon Group Number helps in predicting the biological activity and toxicity of potential drug candidates. For example, drugs with a higher Carbon Group Number may have different pharmacokinetic properties compared to those with a lower Carbon Group Number.
Petroleum Industry
In the petroleum industry, the Carbon Group Number is essential for refining processes. Different hydrocarbons with varying Carbon Group Numbers are separated and processed to produce fuels, lubricants, and other petroleum products. For instance, gasoline typically contains hydrocarbons with Carbon Group Numbers ranging from 5 to 12, while diesel fuel contains hydrocarbons with Carbon Group Numbers ranging from 12 to 20.
Environmental Science
In environmental science, the Carbon Group Number is used to study the behavior and fate of organic pollutants. Understanding the Carbon Group Number helps in predicting the persistence, bioaccumulation, and toxicity of pollutants in the environment. For example, compounds with higher Carbon Group Numbers tend to be more persistent and bioaccumulative, posing greater environmental risks.
Food Science
In food science, the Carbon Group Number is important for understanding the composition and properties of food components. For instance, the Carbon Group Number of fatty acids in oils and fats influences their melting points, stability, and nutritional value. Knowing the Carbon Group Number helps in formulating food products with desired properties and nutritional benefits.
Challenges and Limitations
While the Carbon Group Number is a valuable concept, it also has its challenges and limitations. Some of the key challenges include:
Complex Molecular Structures
For complex molecular structures, determining the Carbon Group Number can be challenging. Spectroscopic techniques and mass spectrometry are often required to accurately determine the number of carbon atoms in such molecules.
Isomers
Isomers are compounds with the same molecular formula but different structural arrangements. Determining the Carbon Group Number for isomers can be complex, as the number of carbon atoms is the same, but the properties and behavior may differ significantly.
Functional Groups
The presence of functional groups can also complicate the determination of the Carbon Group Number. Functional groups, such as hydroxyl, carbonyl, and amino groups, can affect the properties and behavior of the compound, making it difficult to predict based solely on the Carbon Group Number.
Despite these challenges, the Carbon Group Number remains a fundamental concept in organic chemistry, providing valuable insights into the properties and behavior of organic compounds.
In summary, the Carbon Group Number is a crucial concept in organic chemistry that helps in classifying, studying, and understanding organic compounds. It plays a pivotal role in determining the molecular structure, physical properties, chemical reactivity, and applications of various organic compounds. By understanding the Carbon Group Number, chemists can gain deeper insights into the behavior and potential uses of organic compounds, contributing to advancements in various fields, including pharmaceuticals, petroleum, environmental science, and food science. The challenges and limitations associated with the Carbon Group Number highlight the need for advanced analytical techniques and a comprehensive understanding of molecular structures and properties.
Related Terms:
- carbon family
- carbon group number pt
- carbon group elements
- carbon group number periodic table
- carbon group properties
- carbon group periodic table