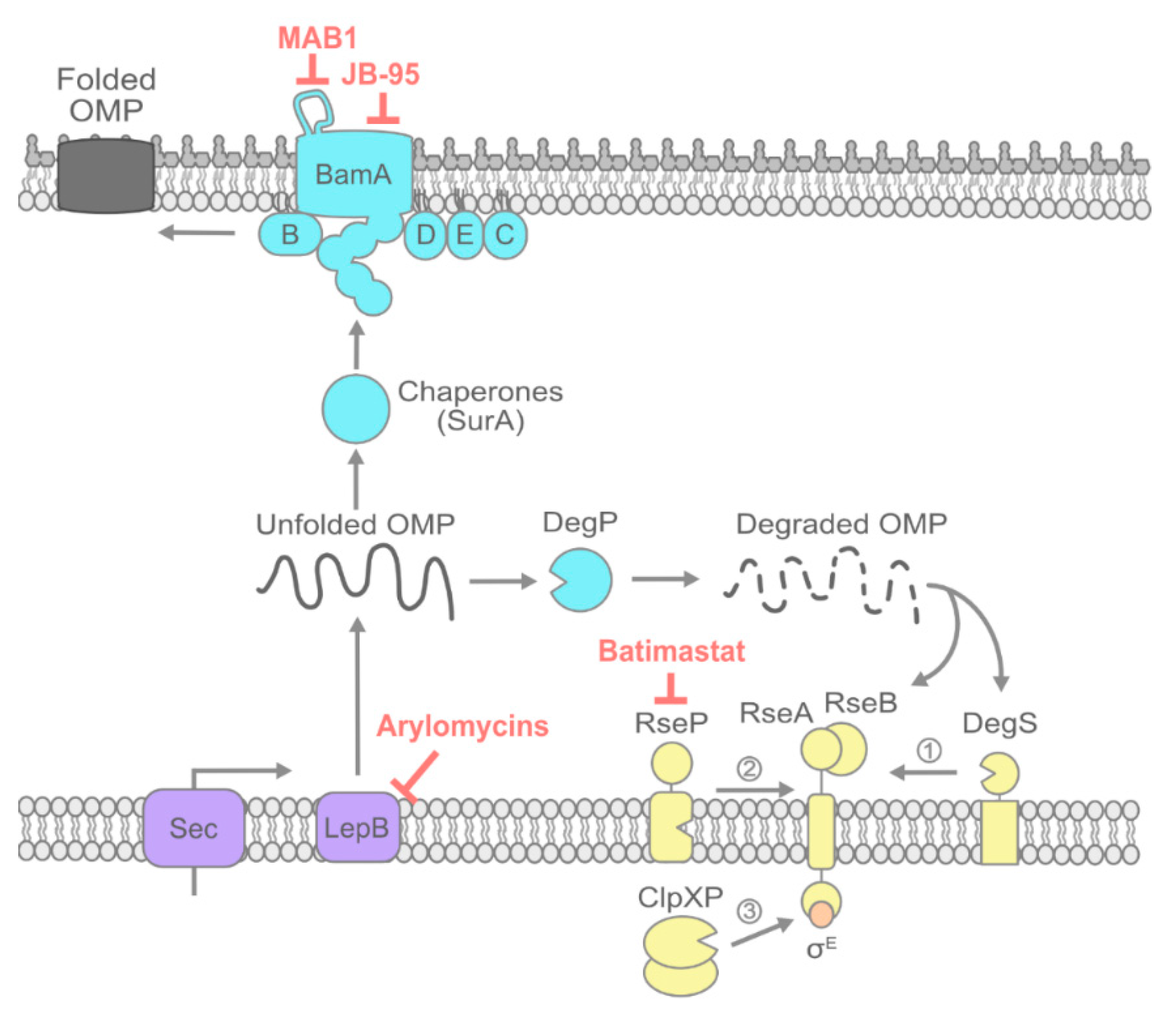
In the realm of microbiology and biochemistry, the study of membrane proteins, particularly those found in bacterial outer membranes, has garnered significant attention. Among these proteins, Are Lptd Porins stand out due to their crucial role in the transport of lipopolysaccharides (LPS) across the outer membrane. This process is essential for the integrity and functionality of the bacterial cell wall, making Lptd Porins a subject of intense research.
Understanding Lptd Porins
Lptd Porins are integral membrane proteins that facilitate the transport of LPS from the inner membrane to the outer membrane of Gram-negative bacteria. LPS is a critical component of the bacterial outer membrane, providing structural support and protecting the cell from environmental stressors. The transport of LPS is a complex process that involves several proteins, with Lptd Porins playing a pivotal role.
The Role of Lptd Porins in LPS Transport
LPS transport is a multi-step process that begins with the synthesis of LPS in the inner membrane. The newly synthesized LPS is then transported across the periplasmic space to the outer membrane, where it is integrated into the membrane structure. Lptd Porins are involved in the final stages of this transport process, acting as a bridge between the periplasmic space and the outer membrane.
The transport mechanism involves several key steps:
- Synthesis of LPS: LPS is synthesized in the inner membrane by a series of enzymatic reactions.
- Transport Across the Periplasmic Space: The LPS is then transported across the periplasmic space by a series of proteins, including LptA, LptC, and LptE.
- Integration into the Outer Membrane: Finally, Lptd Porins facilitate the integration of LPS into the outer membrane, ensuring the structural integrity of the bacterial cell wall.
Structure and Function of Lptd Porins
Lptd Porins are characterized by their unique structure, which allows them to perform their specialized function. These proteins are composed of several transmembrane domains that span the outer membrane, creating a channel through which LPS can pass. The structure of Lptd Porins is highly conserved across different bacterial species, highlighting their essential role in bacterial physiology.
The function of Lptd Porins is closely linked to their structure. The transmembrane domains form a hydrophobic environment that stabilizes the LPS molecules as they pass through the channel. This stabilization is crucial for preventing the aggregation of LPS, which could disrupt the transport process and compromise the integrity of the outer membrane.
Importance of Lptd Porins in Bacterial Pathogenesis
The role of Lptd Porins in LPS transport makes them an important target for antimicrobial therapies. By disrupting the function of Lptd Porins, it is possible to inhibit the transport of LPS, leading to the destabilization of the outer membrane and the eventual death of the bacterial cell. This approach has the potential to overcome the challenges posed by antibiotic resistance, which is a growing concern in the field of infectious diseases.
Research into Lptd Porins has revealed several potential targets for antimicrobial therapies:
- Inhibitors of LPS Transport: Compounds that inhibit the function of Lptd Porins can prevent the transport of LPS, leading to the destabilization of the outer membrane.
- Disruptors of Lptd Porin Structure: Agents that disrupt the structure of Lptd Porins can interfere with their ability to transport LPS, providing a novel approach to antimicrobial therapy.
- Modulators of Lptd Porin Function: Compounds that modulate the function of Lptd Porins can alter the transport of LPS, providing a more targeted approach to antimicrobial therapy.
Challenges and Future Directions
Despite the progress made in understanding the role of Lptd Porins in LPS transport, several challenges remain. One of the primary challenges is the development of effective inhibitors that can specifically target Lptd Porins without affecting other cellular processes. This requires a deep understanding of the structure and function of Lptd Porins, as well as the development of advanced screening techniques to identify potential inhibitors.
Another challenge is the potential for the development of resistance to Lptd Porin inhibitors. Bacteria have a remarkable ability to adapt to new environmental stressors, and it is possible that they could develop mechanisms to circumvent the effects of Lptd Porin inhibitors. To address this challenge, it is essential to develop a diverse range of inhibitors that target different aspects of Lptd Porin function.
Future research in this area will focus on several key areas:
- Structural Studies: Detailed structural studies of Lptd Porins will provide insights into their function and identify potential targets for inhibition.
- Functional Studies: Functional studies will help to understand the mechanisms by which Lptd Porins transport LPS and identify key regulatory steps that can be targeted for inhibition.
- Drug Development: The development of novel inhibitors that specifically target Lptd Porins will be a major focus of future research, with the goal of overcoming the challenges posed by antibiotic resistance.
🔍 Note: The development of effective inhibitors for Lptd Porins requires a multidisciplinary approach, involving expertise in structural biology, biochemistry, and pharmacology.
Comparative Analysis of Lptd Porins Across Different Bacterial Species
Lptd Porins are found in a wide range of Gram-negative bacteria, and their structure and function are highly conserved across different species. However, there are also species-specific differences that can influence their role in LPS transport and bacterial pathogenesis. A comparative analysis of Lptd Porins across different bacterial species can provide valuable insights into their function and identify potential targets for antimicrobial therapies.
| Bacterial Species | Lptd Porin Structure | LPS Transport Mechanism | Potential Targets for Inhibition |
|---|---|---|---|
| Escherichia coli | Highly conserved structure with multiple transmembrane domains | Efficient transport of LPS across the periplasmic space | Inhibitors of LPS transport, disruptors of Lptd Porin structure |
| Pseudomonas aeruginosa | Unique structural features that enhance LPS transport | Enhanced transport of LPS, contributing to antibiotic resistance | Modulators of Lptd Porin function, inhibitors of LPS transport |
| Salmonella enterica | Conserved structure with species-specific variations | Efficient transport of LPS, contributing to virulence | Inhibitors of LPS transport, disruptors of Lptd Porin structure |
This comparative analysis highlights the importance of understanding the species-specific differences in Lptd Porins, as these differences can influence their role in LPS transport and bacterial pathogenesis. By identifying potential targets for inhibition, it is possible to develop more effective antimicrobial therapies that can overcome the challenges posed by antibiotic resistance.
Conclusion
The study of Are Lptd Porins has provided valuable insights into the transport of LPS across the outer membrane of Gram-negative bacteria. These proteins play a crucial role in maintaining the integrity of the bacterial cell wall, making them an important target for antimicrobial therapies. Future research in this area will focus on developing effective inhibitors that can specifically target Lptd Porins, providing a novel approach to overcoming the challenges posed by antibiotic resistance. By understanding the structure and function of Lptd Porins, it is possible to develop more effective antimicrobial therapies that can combat the growing threat of infectious diseases.